The epidemiology of COVID-19 in Tshwane District, South Africa, March 2020-June 2020: A cross-sectional study
Mmatswele Hellen Kgatla1,2,3, Alex De Voux1, Makgomo Rebecca Mphaka3, Hetani Mdose1,2, Alfred Musekiwa2, Emelda Ramutshila1, Mpho Moshime3, Lekwetji Komane3, Lazarus Kuonza 1,2
1South African Field Epidemiology Training Programme, National Institute for Communicable Diseases, Johannesburg, South Africa, 2School of Health Systems and Public Health, Faculty of Health Sciences, University of Pretoria, Pretoria, South Africa, 3Expanded Programme on Immunisation, Communicable Disease Control, and Outbreak Response, Tshwane District, Department of Health, Pretoria, South Africa
&Corresponding author
Mmatswele Hellen Kgatla, South African Field Epidemiology Training Programme, National Institute for Communicable Diseases, Johannesburg, South Africa. kgatlahellen@yahoo.com
Introduction:
On 30 January 2020 the World Health Organization declared the coronavirus disease 2019 (COVID-19), a novel respiratory disease first identified in Wuhan City of China, a Public Health Emergency of International Concern (PHEIC). South Africa (SA) recorded its first case of COVID-19 on 5 March 2020. Tshwane, one of the five districts in Gauteng Province, was among the high transmission zones in the province. This paper describes the epidemiology of COVID-19 in Tshwane from March to June 2020, and identifies the factors associated with hospitalisation and mortality.
Methods:
We analysed surveillance data collected during March–June 2020. Data were analysed using descriptive statistics, and multivariable logistic regression analysis was used to determine factors associated with hospitalisation and mortality.
Results:
During March–June 2020, 5,020 laboratory-confirmed COVID-19 cases were reported in Tshwane. Hospitalisation occurred in 4.9% (n=246/5,020) of cases and 2% of cases died (n=103/5,020). Factors associated with hospitalisation included age >60 years (adjusted odds ratio {aOR} 18.70, 95% CI:11.83–29.55, p<0.001), male gender (aOR:1.38, CI:1.10–1.87, p=0.035); and having a pre-existing comorbidity (aOR:2.89, 95% CI:2.09–3.93, p<0.001). Risk factors for mortality were age >60 years (aOR: 47.13, 95% CI:18.33–121.20, p<0.001), male sex (aOR:1.72, 95% Cl:1.10–2.71, p=0.020), and having a pre-existing comorbidity (aOR:5.58, 95% CI:3.52–8.85, p<0.001).
Conclusion:
COVID-19 was less severe among younger cases. Older age, male sex and pre-existing comorbidities were risk factors for both hospitalisation and mortality. Individuals with these characteristics should be prioritized for management of COVID-19 infections and vaccination.
Introduction

A novel strain of coronavirus was identified in Wuhan City, China in December 2019 [1]. The respiratory disease caused by the novel coronavirus was named Coronavirus Disease 2019 (COVID-19). It continued to spread rapidly and was declared a Public Health Emergency of International Concern by the World Health Organization (WHO) on 30 January 2020 [2]. By 30 June 2020, the global number of infections had surpassed 10 million with over 500,000 deaths [3].
Outbreaks of coronaviruses were reported in the past. In 2003, an outbreak of a novel coronavirus named Severe Acute Respiratory Syndrome (SARS-CoV) was reported [
4]. The outbreak affected 26 countries with 8,096 laboratory-confirmed cases and 774 fatalities. In 2012 another new strain of coronaviruses, now known as Middle East respiratory syndrome coronavirus (MERS–CoV), caused an outbreak that spread across 26 countries [
5]. Patients in these two outbreaks presented with fever, headache, malaise, myalgia, shivering and diarrhoea. These symptoms are similar to COVID-19 symptoms.
South Africa (SA) reported its first case of COVID-19 on 5 March 2020 in a group of travellers returning from Italy [
6]. Rapid spread occurred with 144,264 cases and 2,529 fatalities reported by 30 June 2020 [
3]. SA had the highest number of COVID-19 cases in Africa and was in the top 5 countries with the highest infections globally by 30 June 2020 [
3].
Covid-19 is a highly contagious disease [
7]. According to literature, COVID-19 is transmitted from person to person through droplets, aerosols, sneezing, coughing and fomites2. The COVID-19 droplets can survive on surfaces for many hours. Poor hand hygiene can result in the transmission of these droplets from surfaces to susceptible host [
8]. Although everyone is at risk of COVID-19 infection, some individuals have an increased risk of developing severe disease after infection. Severe COVID-19 infection can result in hospitalisation and mortality [
9]. The elderly, males, smokers and people with a pre-existing co-morbidity were reported to have an increased risk of severe COVID-19 infection, resulting in hospitalisation and death [
10]. In Italy, a case fatality rate of 20.7% was observed in case-patients aged 80 years and above, increasing to 22.7% in those aged 90 years and above [
11]. Similar age-related trends were observed in China, where the median age of deceased cases was 68 years [
10]. In both studies more males died compared to females [
10-11] A high proportion of cases with severe COVID-19 infection reported in global studies had a pre-existing co-morbidity [
12-13] , commonly hypertension, cardiovascular disease, diabetes mellitus and obesity [
12,
14-15],. Smoking was identified as a risk factor for severe COVID-19 disease [
13,
16]. The odds of smoking were 2 times higher in hospitalised, compared to non-hospitalised cases, in Atlanta [
12]. An increase in mortality and hospitalisation of COVID-19 cases resulted in some countries introducing lockdown measures to contain the spread of infection and to relieve pressure on the health systems [
17].
There is a paucity of published studies on COVID-19 in the Sub-Saharan African region and South African context. Tshwane District in South Africa was among the districts with the highest number of COVID-19 cases in the country in June 2020, experiencing an increase in hospitalizations and deaths. With COVID-19 being new, no study has been conducted to determine the predictors of COVID-19 hospitalisation and mortality in Tshwane District, South Africa. Our study aimed to provide evidence on the factors associated with severe COVID-19 infections in Tshwane District, thus contributing to the body of knowledge on this phenomenon.
Methods


Study setting
The study was conducted in Tshwane District, Gauteng Province, which is one of eight metropolitan municipalities in South Africa [
18]. The district is divided into seven regions or sub-districts. It has 72 clinics and 9 hospitals served by the Department of Health, 25 facilities served by the Local Authority, and 62 private health care providers.
Study design and period
We conducted a cross sectional study of COVID-19 laboratory-confirmed cases from March-June 2020.
Study population and selection
Our study population included laboratory-confirmed COVID-19 cases reported to the Tshwane District surveillance system by health care providers and cases received from the National Institute for Communicable Diseases (NICD). We included all the laboratory-confirmed cases, but excluded cases listed as untraceable during contact tracing due to incorrect contact numbers and residential addresses. Criteria for a confirmed case was any person, irrespective of clinical signs and symptoms, with a positive laboratory-conducted polymerase chain reaction (PCR) test result for COVID-19 in Tshwane District, March 2020-June 2020.
Data sources
The data was received from the NICD and those reported through the Tshwane District surveillance system by health care providers. NICD receives data on confirmed COVID-19 cases from both private and public laboratories, allocates the cases into respective provinces. Provincial office then allocates the cases into respective district. Additionally, the district surveillance officer receives clinical notifications of cases from health care providers. We used data from both surveillance systems to ensure that all reported cases were traced.
Data collection
A secondary analysis of surveillance data was conducted. During the study period, any person with COVID-19 defining symptoms or history of contact with a laboratory-confirmed COVID-19 case was considered for testing. Information of any suspected case was reported by health facilities using a standard Person Under Investigation (PUI) form. The district also received a line list of laboratory-confirmed COVID-19 cases from the NICD (which mostly contained the age, gender, residential address and contact details). Due to missing data from the line list provided by the NICD, cases were contacted telephonically to obtain additional information on comorbidities, symptoms and names of people they were in contact with, and to verify their residential address. This information was collated into a line list of only laboratory-confirmed COVID-19 cases.
Study variables
COVID-19 confirmed case
A confirmed case was any person with a positive polymerase chain reaction (PCR) COVID-19 test result irrespective of clinical signs and symptoms.
Asymptomatic case
A COVID-19 laboratory confirmed case without COVID-19 defining symptoms 14 days prior or on the day of testing
Comorbidity
Comorbidity was defined as the presence of one or more medical conditions in cases before COVID-19 infection reported by the cases.
Obesity
Obesity was defined as a body mass index (BMI) of over 30kg/m2
Data analysis
Data were received in a Microsoft Excel 2016 spreadsheet. Duplicates were removed through probabilistic matching using name, surname, age, gender, date of specimen collection, and testing laboratory. An observation was removed if all variables matched. Our data were exported to Stata (Version 15. StataCorp LLC, College Station, TX, United States of America, 2017) for analysis. Data were summarized using descriptive statistics. The mean and standard deviation were used to describe the age of cases. We used graphs and tables to display demographic and clinical presentation of cases. We used univariate and multivariable logistic regression models to identify factors associated with a severe form of infection that required hospitalisation; as well as factors associated with mortality. We collapsed our age groups in the logistic regression model due to the low frequencies seen in hospitalisation and deaths. Factors with a p-value ≤ 0.2 in the univariate analysis were considered for inclusion in the multivariable model using a manual backward elimination procedure. A p-value < 0.05 was considered statistically significant.
Ethical considerations
Ethical approval with waiver of informed consent was granted by the Faculty of Health Sciences Research Ethics Committee of the University of Pretoria (Ethics no: 553/2020) and the Tshwane District Ethics Committee (Ethics no: GP_202010_011).
Results


The epidemiologic characteristics of cases
A total of 5,124 cases were extracted for the period March to June 2020 and 104 were excluded due to an unverified residential address at the time of tracing. The total number included in the study was 5,020 and the mean age was 39.7 (standard deviation (SD) ±16.1) years. The epidemic curve depicts that fewer than 100 cases were diagnosed in the first 11 weeks. An increase in cases was observed from week 12 (end of May 2020)
Figure 1.
The highest number of cases was among those aged 31-40 years (26.2%, 1,315/5,020) with children 10 years and younger having the lowest number of cases (3.5%, 174/5,020). There were more female cases compared to male cases in all the age groups except for those aged 6-10 years
Table 1.
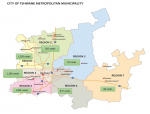
Region 1 (26.5%, 1,328/5,020) and region 3 (25.8%=1,296/5,020) had the highest number of cases. The least number of cases were reported in region 7 (1.3%, 64/5,020) and 5 (1.2%, 61/5,020)
Figure 2.
The clinical presentation of COVID-19 cases
Of the 5,020 cases, 2,192 cases were symptomatic. The most common symptoms were cough (47.1%, 1,033/2,192, sore throat (30.7%, 674/2,192), fever (26.1%, 573/2,192) and headache (26.0%, 571/2,192). Loss of taste and smell were also reported in 13.0%( 286/2,192) and 11.6% (255/2,192) cases respectively.
A total of 567 (11.3%, 567/5,020) cases presented with one or more pre-existing comorbidities. The majority presented with diabetes mellitus (64.5%, 366/567), hypertension (61%, 346/567) and HIV (26.8%, 152/567)
Figure 3. The least commonly reported comorbidity was TB (1%, 5/567).
Death and hospitalisation of severe COVID-19 cases
The highest proportion of cases were hospitalised in April (12.0%, 14/117). Although month of June had the lowest hospitalisation proportion (4.3%, 194/4,535), the proportion of deaths were the highest (2.2%, 98/4,535). The fatality proportion increased monthly after March 2020
Figure 4.
Death and hospitalisation in COVID-19 cases with a pre-existing comorbidities
The highest proportion of deaths (50%, 6/12) occurred in cases who were obese. Half of these cases were hospitalised and they all subsequently died. The second -highest proportion of cases hospitalised (53.3%, 8/15) was reported among cases with a kidney problem, with a fatality proportion of 46.7% (7/15). Three out of five TB cases with COVID-19 were hospitalised, but no death was reported among these cases
Figure 3.
Clinical characteristics of hospitalised cases
A total of 246 (4.9%) cases were hospitalised. The number of hospitalised cases increased with age and more than half were males aged 60 years and above (52.4%,129/246). Most of these cases were over the age of 50 years (65.9%, 162/246) and 47.2% (n=116/246) had no pre-existing comorbidity
Table 2.
Factors associated with severe COVID-19 infection requiring hospitalisation
In our univariate analysis, we included age, gender and having pre-existing comorbidities as predictors for severe infection that resulted in hospitalisation. Males had almost 1.5 times higher odds of hospital admission compared to females (OR:1.47, 95%CI:1.14-1.90, p=0.003). Cases aged over 60 years had 23 times increased odds of hospitalisation than those aged 40 years and younger (OR 23.09, 95%CI: 15.08-35.13, p<0.001). The odds of hospital admission were higher among those with a pre-existing comorbidity (OR 4.32, 95%CI: 3.22-5.79, p<0.001).
When these variables were included in a multivariable logistic regression model older age, being male and having a pre-existing comorbidity were independent predictors of COVID-19 hospitalization. The odds of having severe COVID-19 infection increased with an increase age. The highest odds were observed in those aged 60 years and older (aOR: 18.70, 95%CI:11.83-29.60, p<0.001). The odds of hospital admission were 1.4 times higher in males than in females (aOR 1.38, 95%CI:1.10-1.87, p=0.035) The odds of admission were two times higher in those with pre-existing comorbidities than those without (aOR 2.90, 95%CI: 2.10-3.93, p<0.001)
Table 3.
Factors associated with COVID-19 mortality in Tshwane District, March- June 2020
We included age, gender and pre-existing comorbidity as predictors for death due to COVID-19 in the univariate analysis. Those aged > 60 years had 71 times increased odds of dying from COVID-19 than those aged 40 years and younger (OR 71.3, 95%CI: 28.33-179.46, p<0.001). Males had increased odds of dying from COVID-19 compared to females (OR 1.77, 95%CI: 1.20-2.64, p=0.004). Having pre-existing comorbidities increased the odds of death (OR 9.00, 95%CI:5.80-13.92, p<0.001) compared to COVID-19 cases without any pre-existing comorbidities. Risk factors associated with COVID-19 mortality were age, gender and pre-existing co-morbidity. The odds of dying were 47 times higher in cases aged over 60 years compared to those aged 40 years and younger (aOR 47.13, 95%CI: 13.32-121.20, p <0.001). This wide confidence interval might be due to a small number of cases among the elderly. The odds of mortality were 1.72 times higher in males compared to females (aOR 1.72, 95%CI:1.10-2.72, p=0.020). Having pre-existing comorbidities increased the odds of COVID-19 mortality (aOR 5.58, 95%CI: 3.52-8.85, p<0.001). The goodness of fit of the multivariate model was adequate (p=0.1891)
Figure 4.
Discussion


Our study aimed to describe the epidemiology of COVID-19 cases in Tshwane District, South Africa, during the period from March to June 2020. We observed an increase in cases from end of May to end of June 2020. We also aimed to determine factors that were associated with a severe form of infection that resulted in hospitalisation and deaths. Our study found that older age, male gender, and having a pre-existing comorbidity was associated with a severe form of infection resulting in hospitalisation. Risk factors associated with mortality were older age, male gender and having a pre-existing comorbidity.
The increase in the number of COVID-19 cases in June 2020 is comparable with the increase reported in Gauteng Province, during which period the province was the epicentre of the pandemic in the country. The national testing capacity was increased between March and June 2020 [
19]. In the same period, South Africa´s positivity rate increased from 2.2% at the beginning of March to 25.8% towards the end of June 2020. Gauteng conducted the highest number of tests compared to other provinces [
19]. South Africa relaxed its lockdown measures from 1 June 2020, increasing economic activities, reopening of schools and allowing the sale of alcohol. This could have contributed to the increase in COVID-19 cases [
20]. Alcohol consumption increased the spread of the virus through the sharing of beverage glasses and loss of ability to adhere to non-pharmaceutical prevention interventions such as physical distancing, regular handwashing and wearing of masks [
21].
The demographic characteristics of COVID-19 cases have been studied globally, with age, gender and pre-existing comorbidity as predictors of mortality. The elderly has been found to be at an increased risk of a severe form of infection that required hospitalisation [
9,
22]. Age was found to be a predictor of SARS and MERS mortality in past outbreaks [
23]. The elderly people have a deteriorating immune system, which can affect their ability to fight infections. Men were at a higher risk of death as compared to women. Our findings were similar to studies conducted in New York City, USA and Wuhan City, China [
9,
24]. Differences in lifestyle, genetics and health seeking behaviour between males and females may contribute to the significant difference in susceptibility [
25]. Although not collected in this study, it is known that males are more likely to smoke, which could predispose them to severe infection [
16,
22].
COVID-19 cases can present as symptomatic or asymptomatic. Cough, sore throat fever and headache were the most common COVID-19 symptoms recorded in our study. Headache was a less common symptom in most studies [
26-27]. Studies have reported loss of smell and taste as predictors of COVID-19 infection [
28-29]. Our study also identified an increasing number of cases presenting with these symptoms. Loss of smell and taste often occur in 2.3% of influenza cases [
30].
The presence of a pre-existing comorbidity has been identified as a risk factor for COVID-19 hospitalisation and death [
9,
14,
22] Comorbidities impair the immune system, affecting an individual´s ability to fight infections [
31]. Similar trends were observed in the MERS and influenza A virus (H1N1) outbreaks [
30-33]. Obesity and kidney disease were common in hospitalised and deceased cases in our study. This is in contrast to the findings in Italy where chronic obstructive pulmonary disease and hypercholesterolemia were common in cases with a severe form of COVID-19 infection [
14] An increase in the form of infection resulting in hospitalisation and death of obese cases was reported in studies in China, the USA and by WHO [
9,
32]. People with obesity are at a higher risk of chronic diseases such as diabetes mellitus, asthma, cardiovascular disease and hypertension [
22]. In the twelve cases who were recorded as obese in our study, nine had a co-morbidity such as diabetes, heart conditions and hypertension. This might have contributed to the increased risk of hospitalisation and death in obese COVID-19 cases [
22]. Obesity leads to difficulty in breathing even in the absence of COVID-19; increasing airway resistance, decreasing expiratory reserve volume, functional capacity and pulmonary compliance. Although HIV was among the top reported comorbidities, the proportion of hospitalisations and deaths among HIV-positive individuals was less than 10%. The low proportion in hospitalisation and deaths could be due to the lower prevalence of HIV in older age groups. Of the 152 COVID-19 cases with HIV, only 4 were aged over 60 years. The same trend was reported in the MERS outbreak with less than 5% of HIV positive cases developing severe infection [
31].
The district´s mortality proportion was similar to South Africa´s national COVID-19 mortality proportion of 2%; but lower than the global mortality rate of 4.9% reported on 30 June 2020 [
3]. The African region also reported a 2% mortality proportion [
3]. In contrast, Wuhan and Italy reported mortality rates of 3.6% and 7.2%, respectively [
11,
34]. The difference in COVID-19 mortality between countries may be due to variation in the demographic characteristics of their populations. South Africa has a higher proportion of younger generation inhabitants, compared to Italy that has 23% of its population over the age of 65 years [
11].
Our study used data reported from all facilities in the Tshwane District, therefore the data were representative of cases in the district. It however had a number of limitations. Firstly, we had missing information on cases, resulting in exclusion of some. Secondly, information of the oxygen status, treatment and length of hospitalisation, which were identified as predictors of mortality in other studies, were missing [
10-11]. Therefore, we could not conduct an in-depth analysis of the hospitalised cases. Thirdly, our deaths were only facility-based deaths and those identified during contact tracing, so any death that could have occurred outside of the health facility, or occurred after contact tracing would have been missed. We acknowledge that their clinical characteristics could be different from the deaths reported in our study. It is also possible that there was misclassification bias for symptoms as some of the cases might have developed symptoms after testing was done. We also acknowledge potential misclassification and underreporting of comorbidities as it was self-reported.
Conclusion


The epidemiological characteristics of cases in our study were consistent with global studies. Age, gender and pre-existing comorbidities were important predictors of COVD-19 severity that resulted in hospitalisation and death. These findings are important in focusing strategies in the prevention of severe or complicated COVID-19 infections and in policy formulations. We recommend that the elderly, males and people with a pre-existing comorbidity should be at the centre of prevention strategies and prioritized when vaccines are developed and rolled-out. There is a need for a follow-up study to provide a more comprehensive analysis of severe COVID-19 cases looking at clinical records of these cases.
What is known about this topic
- COVID-19 is a pandemic of public health concern
- South Africa has the highest COVID-19 cases in the WHO African region
- The pandemic continues to spread and studies are conducted worldwide to understand the epidemiology of the virus
What this study adds
- COVID-19 is a pandemic of public health concern
- South Africa has the highest COVID-19 cases in the WHO African region
- The pandemic continues to spread and studies are conducted worldwide to understand the epidemiology of the virus
Competing interests


No conflict of interest was declared.
Funding
The study was funded by the South African Field Epidemiology Training Programme (SAFETP).
Authors´ contributions


KH, DA, MH MM, ER and MR conceived the study. KH carried out the ethical application, analysis, and drafted the first manuscript. MA guided the analysis. KH, DA, MA, MM, MR, KL, RE and KL revised the manuscript and gave authorisation for publication.
Acknowledgments


We acknowledge Tshwane District COVID-19 tracers team for their contribution in collecting the data used for the study and the management for permission to use the data. We acknowledge the supervisors from Tshwane District Department of Health and the South African Field Epidemiology Programme for their constant availability in reviewing the paper.
Tables and figures


Table 1: Clinical characteristics of hospitalised COVID-19 cases, Tshwane District, South Africa, March-June 2020
Table 2: Factors associated with hospitalisation in COVID-19 cases, Tshwane District, South Africa, March-June 2020
Table 3: Factors associated with mortality in COVID-19 cases, Tshwane District, South Africa, March-June 2020
Figure 1: Epidemiological curve of COVID-19 cases in Tshwane District, March-June 2020
Figure 2: The distribution of COVID-19 cases in Tshwane District by regions, March-June 2020
Figure 3: Number of confirmed COVID-19 cases by pre-existing comorbidities, March-June 2020
Figure 4: Proportion of COVID-19 case hospitalisation and deaths by month in Tshwane District, March-June 2020
References


- Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med[internet]. 2020 Feb 20[cited 2023 Feb 8]; 382(8):727-33.https://doi.org/10.1056/nejmoa2001017 . Google Scholar
- Sohrabi C, Alsafi Z, O'Neill N, Khan M, Kerwan A, Al-Jabir A, Iosifidis C, Agha R. World Health Organization declares global emergency: A review of the 2019 novel coronavirus (COVID-19). Int J Surg[Internet]. 2020 Apr[cited 2023 Feb 8]; 76:71-76.https://doi.org/10.1016/j.ijsu.2020.02.034 PubMed | Google Scholar
- WHO. Coronavirus disease (COVID-19) Situation Report - 162[Internet]. World Health Organization; 2020 Jun[cited 2023 Feb 8]. Report No.: 162.
- WHO. Severe Acute Respiratory Syndrome (SARS)[Internet]. Word Health Organization; 2004 March 24 [cited 2023 Feb 8].
- Bennet N. Alarm bells over MERS coronavirus. Lancet Infect Dis[Internet]. 2013 Jul[cited 2023 Feb 8]; 13(7):573-4. https://doi.org/10.1016/s1473-3099(13)70135-x PubMed | Google Scholar
- National Institute for Communicable Diseases. First Case of Covid-19 Coronavirus Reported in SA[Internet]. NICD; 2020 March 5[cited 2021 Sep 8].
- Lotfi M, Hamblin MR, Rezaei N. COVID-19: Transmission, prevention, and potential therapeutic opportunities. Clin Chim Acta[Internet]. 2020 Sep[cited 2023 Feb 9]; 508:254-266.https://doi.org/10.1016/j.cca.2020.05.044 PubMed | Google Scholar
- Jayaweera M, Perera H, Gunawardana B, Manatunge J. Transmission of COVID-19 virus by droplets and aerosols: A critical review on the unresolved dichotomy. Environ Res[Internet]. 2020 Sep[cited 2023 Feb 9]; 188:109819.https://doi.org/10.1016/j.envres.2020.109819 PubMed | Google Scholar
- Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW, and the Northwell COVID-19 Research Consortium, Barnaby DP, Becker LB, Chelico JD, Cohen SL, Cookingham J, Coppa K, Diefenbach MA, Dominello AJ, Duer-Hefele J, Falzon L, Gitlin J, Hajizadeh N, Harvin TG, Hirschwerk DA, Kim EJ, Kozel ZM, Marrast LM, Mogavero JN, Osorio GA, Qiu M, Zanos TP. Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized with COVID-19 in the New York City Area. JAMA[Internet]. 2020 May 26[cited 2023 Feb 9]; 323(20):2052-205. https://doi.org/10.1001/jama.2020.6775. Google Scholar
- Chen T, Wu D, Chen H, Yan W, Yang D, Chen G, Ma K, Xu D, Yu H, Wang H, Wang T, Guo W, Chen J, Ding C, Zhang X, Huang J, Han M, Li S, Luo X, Zhao J, Ning Q. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ[Internet]. 2020 Mar 31[cited 2023 Feb 8]; 368:m1295. Erratum in BMJ. 2020 Mar 31;368:m1295. https://doi.org/1136/bmj.m1091. Google Scholar
- Onder G, Rezza G, Brusaferro S. Case-fatality rate and characteristics of patients dying in relation to COVID-19 in Italy. JAMA[Internet]. 2020 May 12[cited 2023 Feb 21]; 323(18):1775-6.https://doi.org/10.1001/jama.2020.4683. Google Scholar
- Killerby ME, Link-Gelles R, Haight SC, Schrodt CA, England L, Gomes DJ, Shamout M, Pettrone K, O'Laughlin K, Kimball A, Blau EF, Burnett E, Ladva CN, Szablewski CM, Tobin-D'Angelo M, Oosmanally N, Drenzek C, Murphy DJ, Blum JM, Hollberg J, Lefkove B, Brown FW, Shimabukuro T, Midgley CM, Tate JE; CDC COVID-19 Response Clinical Team. Characteristics Associated with Hospitalization Among Patients with COVID-19 - Metropolitan Atlanta, Georgia, March-April 2020. MMWR Morb Mortal Wkly Rep[Internet]. 2020 Jun 26[cited 2023 Feb 9]; 69(25):790-794. https://doi.org/10.15585/mmwr.mm6925e1 PubMed | Google Scholar
- Zheng Z, Peng F, Xu B, Zhao J, Liu H, Peng J, Li Q, Jiang C, Zhou Y, Liu S, Ye C, Zhang P, Xing Y, Guo H, Tang W. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. J Infect[Internet]. 2020 Aug[cited 2023 Feb 9]; 81(2):e16-e25. https://doi.org/10.1016/j.jinf.2020.04.021 PubMed | Google Scholar
- Grasselli G, Greco M, Zanella A, Albano G, Antonelli M, Bellani G, Bonanomi E, Cabrini L, Carlesso E, Castelli G, Cattaneo S, Cereda D, Colombo S, Coluccello A, Crescini G, Forastieri Molinari A, Foti G, Fumagalli R, Iotti GA, Langer T, Latronico N, Lorini FL, Mojoli F, Natalini G, Pessina CM, Ranieri VM, Rech R, Scudeller L, Rosano A, Storti E, Thompson BT, Tirani M, Villani PG, Pesenti A, Cecconi M; COVID-19 Lombardy ICU Network. Risk Factors Associated With Mortality Among Patients With COVID-19 in Intensive Care Units in Lombardy, Italy. JAMA Intern Med[Internet]. 2020 Oct 1[cited 2023 Mar 2];180(10):1345-1355. Erratum in: JAMA Intern Med. 2021 Jul 1; 181(7):1021. https://doi.org/10.1001/jamainternmed.2020.3539. PubMed | Google Scholar
- Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, Guan L, Wei Y, Li H, Wu X, Xu J, Tu S, Zhang Y, Chen H, Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The Lancet[Internet]. 2020 Mar 28[cited 2023 Feb 9]; 395(10229):1054-1062.https://doi.org/10.1016/S0140-6736(20)30566-3 PubMed | Google Scholar
- Cai H. Sex difference and smoking predisposition in patients with COVID-19. The Lancet Respiratory Medicine[Internet]. 2020 Apr 1[cited 2023 Feb 9]; 8(4):e20. https://doi.org/10.1016/s2213-2600(20)30117-x. Google Scholar
- Hamzelou J. World in lockdown. New Scientist[Internet]. 2020 Mar[cited 2023 Feb 9]; 245(3275):7. https://doi.org/10.1016/S0262-4079(20)30611-4. Google Scholar
- Government of South Africa. Local government[Internet]. Government of South Africa.[cited 2023 Feb 9].
- National Institute for Communicable Diseases (NICD). Covid-19 Testing Summary-South Africa: Week 34 2020[Internet]. NICD; 2020[cited 2023 Feb 8]. 22 p.
- National Department of Health, Republic of South Africa. National Lockdown Regulations - SA Corona Virus Online Portal[Internet]. SA Corona Virus Online Portal. 2020 Mar 26[cited 2020 Apr 27].
- Mungmungpuntipantip R, Wiwanitkit V. Sharing Alcoholic Drinks and a COVID-19 Outbreak. Alcohol and Alcoholism[Internet]. 2020 Jun 25[cited 2023 Feb 9]; 55(4):343. https://doi.org/10.1093/alcalc/agaa028 PubMed | Google Scholar
- Palaiodimos L, Kokkinidis DG, Li W, Karamanis D, Ognibene J, Arora S, Southern WN, Mantzoros CS. Severe obesity, increasing age and male sex are independently associated with worse in-hospital outcomes, and higher in-hospital mortality, in a cohort of patients with COVID-19 in the Bronx, New York. Metabolism[Internet]. 2020 Jul[cited 2023 Feb 9]; 108:154262. https://doi.org/10.1016/j.metabol.2020.154262 PubMed | Google Scholar
- Lu L, Zhong W, Bian Z, Li Z, Zhang K, Liang B, Zhong Y, Hu M, Lin L, Liu J, Lin X, Huang Y, Jiang J, Yang X, Zhang X, Huang Z. A comparison of mortality-related risk factors of COVID-19, SARS, and MERS: A systematic review and meta-analysis. J Infect[Internet]. 2020 Oct[ cited 2023 Feb 9];81(4):e18-e25. https://doi.org/10.1016/j.jinf.2020.07.002 PubMed | Google Scholar
- Du RH, Liang LR, Yang CQ, Wang W, Cao TZ, Li M, Guo GY, Du J, Zheng CL, Zhu Q, Hu M, Li XY, Peng P, Shi HZ. Predictors of mortality for patients with COVID-19 pneumonia caused by SARS-CoV-2: a prospective cohort study. Eur Respir J[Internet]. 2020 May[cited 2023 Feb 9]; 55(5):20005. https://doi.org/10.1183/13993003.00524-2020 Google Scholar
- Bhopal SS, Bhopal R. Sex differential in COVID-19 mortality varies markedly by age. The Lancet[Internet]. 2020 Aug 22[cited 2023 Feb 9]; 396(10250):532-533. https://doi.org/10.1016/s0140-6736(20)31748-7 PubMed | Google Scholar
- Paules CI, Marston HD, Fauci AS. Coronavirus Infections-More Than Just the Common Cold. JAMA[Internet]. 2020 Feb 25[cited 2023 Feb 9]; 323(8):707-08. https://doi.org/10.1001/jama.2020.0757. Google Scholar
- Bi Q, Wu Y, Mei S, Ye C, Zou X, Zhang Z, Liu X, Wei L, Truelove SA, Zhang T, Gao W, Cheng C, Tang X, Wu X, Wu Y, Sun B, Huang S, Sun Y, Zhang J, Ma T, Lessler J, Feng T. Transmission of COVID-19 in Shenzhen China: Analysis of 391 cases and 1,286 of their close contactsEpidemiology and . medRxiv [Preprint]. 2020 Mar Published in: The Lancet Infectious Diseases. 2020 Aug 1; 20(8):911-9.https://doi.org/10.1101/2020.03.03.20028423 . Google Scholar
- Menni C, Valdes AM, Freidin MB, Ganesh S, El-Sayed Moustafa JS, Visconti A, Hysi P, Bowyer RCE, Mangino M, Falchi M, Wolf J, Steves CJ, Spector TD. Loss of smell and taste in combination with other symptoms is a strong predictor of COVID-19 infection. medRxiv [Preprint] 2020 April 07. Published in: Nat Med. 2020 Jul; 26(7):1037-40. https://doi.org/10.1101/2020.04.05.20048421. Google Scholar
- Lao WP, Imam SA, Nguyen SA. Anosmia, hyposmia, and dysgeusia as indicators for positive SARS–CoV–2 infection. World j otorhinolaryngol-head neck surg[Internet]. 2020 Nov[cited 2023 Feb 9]; 6(S1).https://doi.org/10.1016/j.wjorl.2020.04.001 . Google Scholar
- de Haro-Licer J, Roura-Moreno J, Vizitiu A, González-Fernández A, González-Ares JA. Long term serious olfactory loss in colds and/or flu. Acta Otorrinolaringologica (English Edition)[Internet]. 2013 Sep[cited 2023 Feb 9]; 64(5):331-8.https://doi.org/10.1016/j.otoeng.2013.10.004 . Google Scholar
- Badawi A, Ryoo SG. Prevalence of comorbidities in the Middle East respiratory syndrome coronavirus (MERS-CoV): a systematic review and meta-analysis. Int J Infect Dis[Internet]. 2016 Aug[cited 2023 Feb 9]; 49:129-33. https://doi.org/10.1016/j.ijid.2016.06.015 PubMed | Google Scholar
- Michalakis K, Ilias I. SARS-CoV-2 infection and obesity: Common inflammatory and metabolic aspects. Diabetes Metab Syndr[Internet]. 2020 Jul[cited 2023 Feb 9]; 14(4):469-471. https://doi.org/10.1016/j.dsx.2020.04.033 PubMed | Google Scholar
- Sun Y, Wang Q, Yang G, Lin C, Zhang Y, Yang P. Weight and prognosis for influenza A(H1n1)pdm09 infection during the pandemic period between 2009 and 2011: a systematic review of observational studies with meta-analysis. Infectious Diseases[Internet]. 2016 Dec 1[cited 2023 Feb 9]; 48(11-12):813-22. https://doi.org/10.1080/23744235.2016.1201721. Google Scholar
- Baud D, Qi X, Nielsen-Saines K, Musso D, Pomar L, Favre G. Real estimates of mortality following COVID-19 infection. The Lancet Infectious Diseases[Internet]. 2020 Jul[cited 2023 Feb 9]; 20(7):773.https://doi.org/10.1016/S1473-3099(20)30195-X .