Prompt Response to a Cross-border Plague Outbreak in Zombo District, Minimized Spread, Uganda, March 2019
Doreen Nsiimire Gonahasa1,&, Bernadette Mirembe Basuta1, Sandra Nabatanzi1, Benon Kwesiga1, Lilian Bulage1, Alex Riolexus Ario1
1Uganda Public Health Fellowship Program, Ministry of Health, Kampala, Uganda
&Corresponding author
Doreen Nsiimire Gonahasa, Uganda Public Health Fellowship Program, Ministry of Health, Kampala, Uganda. dorynegona@musph.ac.ug
Introduction:
Plague, which is caused by the bacterium Yersinia pestis, is a priority zoonotic disease targeted for elimination in Uganda. On 6 March 2019, the Uganda Ministry of Health was notified of a patient in Zombo District with clinical presentation similar to pneumonic plague, and a positive plague rapid diagnostic test (RDT). We determined the scope of the outbreak, determined the mode of transmission, and recommended evidence-based control and prevention measures.
Methods:
A suspected pneumonic plague case was one with two or more of the following signs and symptoms: cough (bloody or wet), chest pain, difficulty in breathing, or fever in a resident of Zombo District during February 1-March 31, 2019. A confirmed case was a suspected case testing positive for Yersinia pestis by rapid diagnostic test, culture or serology. We actively searched for case-patients, traced contacts and took samples as appropriate. We performed descriptive epidemiology of the outbreak.
Results:
We identified one suspected and one confirmed pneumonic plague case. On February 26, 2019, a 4-year-old boy was buried in DRC near the Uganda border after succumbing to bubonic plague. Case-patient A (35-year-old mother to the boy), fell ill with suspected pneumonic plague while attending to him. She was referred to a health facility in Uganda on February 28 but died on arrival. On March 4, Case-patient B (23-year-old sister to Case-patient A), presented with pneumonic plague symptoms to the same Uganda facility and tested plague-positive by RDT, culture, and serological tests. Contacts (n=114) were traced and given prophylaxis; no new cases were reported.
Conclusion:
This fatal plague outbreak started as bubonic and later manifested as pneumonic. There was cross-border spread from DRC to Uganda with no cross-border efforts at prevention and control. Person-to-person transmission appears to have occurred. The quick and effective response likely minimized spread.
Introduction

Plague is a zoonotic disease caused by the bacterium Yersinia pestis and found in wild rodents and their fleas [1]. It is transmitted to human beings when bitten by a rodent flea carrying the bacterium [2]. It may present as bubonic, pneumonic, or septicemic plague [3]. Bubonic plague, which results when an infected flea bites a human or exposure in infected material through broken skin, manifests as swollen nymph nodes called buboes. These may be inflamed and painful and may turn into open sores filled with pus in advanced stages. Pneumonic plague is the most virulent form and may result from progression of untreated bubonic plague or be transmitted by droplets from an infected person [3]. Septicemic plague manifests by darkening of parts of the body caused by infestation of the blood vessels by the bacteria [4]. The incubation period of plague ranges between 1-8 days, depending on the mechanism of infection and subsequent presentation [5]. Untreated plague has a fatality rate of 50-60% [6], but may be as high as 100% in septicemic plague cases [7].
At least three plague pandemics have been documented, each of which have left reservoirs in affected areas, including Europe, Asia, and Africa [
8]. Although sporadic cases have occurred globally in recent years [
9], improved sanitation has limited its spread [
6]. Advancements in diagnostics and access to appropriate antibiotic therapy have also substantially reduced case-fatality rates [
10]. Despite the decrease in human plague cases, plague bacteria continue to circulate in rodent hosts and their fleas in plague-endemic areas, including Madagascar [
11], Democratic Republic of the Congo (DRC) [
12], and northwestern Uganda [
4].
Plague is considered a priority zoonotic disease in Uganda and is targeted for elimination [
13]. It is listed as a disease to be reported immediately under the Integrated Disease Surveillance and Response (IDSR) National Technical Guidelines [
13]. Plague has been documented for decades in West Nile region, in north-western Uganda, as well as the neighbouring Ituri Province in DRC [
12]. West Nile Region is a densely-populated region with a considerable refugee population [
14]. It is located near the borders of DRC and South Sudan and predominantly has a subsistence agricultural economy. Over the years, there have been sporadic cases of plague in this region, with the overall incidence decreasing gradually since 2009 [
4]. The decline in plague cases has likely been due to efforts by the Uganda Virus Research Institute to conduct plague surveillance and community sensitization on prevention methods in the region, including availing of (rapid diagnostic test) RDT kits in the region [
4] enabling health workers to manage suspects appropriately.
On 6 March 2019, The Uganda Ministry of Health Public Health Emergency Operations Center (PHEOC) was notified of an adult female presenting with fever, haemoptysis, and difficulty breathing admitted at Warr Health Center III in Zombo District. The patient tested positive for plague by RDT. It was reported that her family members had travelled during the past week to DRC for burial of a person who had died with similar symptoms. The patient had returned to Uganda with her ailing sister, whom she nursed, and other family members. We investigated to determine the scope of the outbreak and the mode of transmission, and recommended evidence-based control and prevention measures.
Methods


Outbreak area
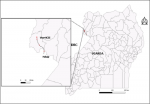
The outbreak occurred in Warr Sub-county, Zombo District. Zombo District is bordered by the DRC, about 20km from Mahagi, Ituri Province where there had been an ongoing plague outbreak
Figure 1. The district is located in the West Nile region in northwestern Uganda, the current plague focus of Uganda.
Case definition and finding
We defined a suspected pneumonic plague case was onset of two or more signs and symptoms of: cough (bloody or wet), chest pain, difficulty breathing, or fever in a resident of Zombo District during February 1-March 31, 2019. A confirmed case was a suspected case testing positive for
Yersinia pestis by rapid diagnostic test, culture or serology.
Between 9th and 17th March, we actively searched for cases within the communities through support by village health teams (VHTs). We also visited three health facilities (Warr HCIII, Kango HCII, and Holy Family Hospital, Nyapea) where we reviewed patient records to identify possible cases of pneumonic plague.
Contact tracing and follow up
We traced both primary and secondary contacts of the suspected case-patient using the World Health Organization (WHO) Integrated Disease Surveillance and Response (IDSR) contact tracing form [
13]. We defined primary contacts as persons who cared for other case-patients or stayed in the same room with them, while secondary contacts were those that stayed in the same household or facility where the case-patients were managed [
15]. Contacts were followed for seven days to observe any symptom development.
Laboratory Investigations
Sputum and whole blood were taken from the single surviving case-patient. From these samples, a plague RDT at Warr Health Centre, culture, and PCR for plague at the CDC laboratory in Atlanta, USA were conducted. Multiple tests were used to help provide rapid results (RDT) and have confidence in results (culture).
Environmental Assessment
Interviews were carried out and included questions on potential risk factors such as type of housing, food storage and domestic rat die offs amongst others in the affected village and subcounty
Ethical Considerations
This investigation was in response to a public health emergency and was therefore determined to be non-research. The Uganda Ministry of Health gave the directive and approval to investigate this outbreak. The Office of the Associate Director for Science at the CDC´s Center for Global Health determined that this activity was not human subjects research and that its primary intent was disease control. Verbal informed consent in the local language was sought from respondents. They were informed that their participation was voluntary, and their refusal would not result in any negative consequences. Contacts who received prophylaxis were informed of its benefits and possible risks.
Results


Chronology of events and case-patients´ description
On February 26, 2019, a 4-year-old Congolese boy was buried in Mahagi Parish of the Ituri Province of DRC, near the Ugandan border where his family lived. On February 28, 2019, Case-patient A, the 35-year-old mother of the boy, also a resident of DRC, was brought to Warr Health Centre III in Zombo District, Uganda, by Ugandan family members. She was febrile (temperature not recorded), coughing blood, and reporting difficulty breathing and chest pain. Her family members reported that she had been caring her ill son in DRC before he died. The family members, who had traveled to DRC to attend the burial of her son, noted that she was ill at the burial and brought her back to Uganda for treatment. Unfortunately, case-patient A died shortly after arrival at the hospital. Healthcare workers did not administer any diagnostic tests. The movement routes of the family from the DRC to Uganda are shown in
Figure 1.
On 4 March, case-patient B, the 23-year-old sister to case-patient A presented at Warr HCIII with headache, fever, chills, sore throat, chest pain, difficulty breathing, coughing blood, malaise, prostration, and anorexia. Case-patient B had cared for case-patient A during her illness on return to Uganda but had not traveled to DRC. An RDT conducted on sputum sample from case-patient B was positive for plague. She was immediately isolated and managed with oral ciprofloxacin until recovery. Further confirmatory tests by culture and PCR were also positive for
Yersinia pestis.
Contact tracing and follow up findings
We identified 114 contacts for both case-patients, including 44 primary contacts and 59 secondary contacts. Eleven (9.6%) of the contacts were healthcare workers. Of the 114 contacts, 65% were female and median age was 40 years (Range: 1-75 years). All contacts were given doxycycline (for primary contacts that came into direct contact with patient body fluids) or cotrimoxazole (for secondary contacts who were in close proximity with the patient). Contacts were followed for seven days and observed for signs and symptoms of plague. No new cases were identified. The nearby communities were sensitized on plague prevention, and healthcare workers at Warr Health Centre were provided with a continuing medical education course on plague.
Environment assessment
Case persons came from a homestead with mud and wattle grass thatched huts with gardens and bushes surrounding the homestead. The case persons both had a primary education and were crop farmers by occupation. Food was kept in the households however there was no history of domestic rat die offs in the past week suggestive of bubonic plague outbreak in the affected village.
Discussion


Our epidemiological investigation revealed a pneumonic plague outbreak in Uganda imported from the DRC. The outbreak represented human-to-human transmission in a single family. Timely response was facilitated by health workers with a high index of suspicion, availability of rapid test kits, and a functional surveillance system that enabled rapid identification and response. This rapid response likely minimized spread.
The Ituri Province of the DRC is endemic for plague, and the province reported additional cases of plague in late 2019 [
16] as well as during June 2020 [
17]. Northwestern Uganda also experiences sporadic plague cases [
4]. Both of these areas lie at similar altitudes (>1300 m), with lower temperatures and higher rainfall than the surrounding lowlands [
18]. Such areas, along with Rift Valley escarpments, have been found to be at elevated risk for plague [
10]. Hotspots of pneumonic plague in Madagascar have also been identified in ecologically similar regions, in the northeastern central highlands, where the median elevation is 1200-1300 m [
19].
Despite sporadic cases of plague in Uganda, the incidence has gradually been decreasing [
4]. This may be in part attributed to an enhanced surveillance program begun in 2003 by the Uganda Virus Research Institute (UVRI). The UVRI Plague Program has been actively involved in surveillance of plague in the West Nile Region with the aim of understanding the plague ecology and epidemiology, developing appropriate diagnostics, patient treatment and care guidelines, and effective public health interventions. This program has also included sensitization of communities about plague, which may have led to greater community awareness and prevention measures [
4]. Additionally, health worker trainings through Continuous Medical Education (CMEs) has facilitated early warning and detection. According to the WHO, such a comprehensive surveillance system is necessary for disease targeted for elimination or eradication, including immediate reporting [
20] The immediate reporting and timely investigation and adoption of interventions therefore minimised the magnitude of the outbreak.
Case-patient A likely died as a result of delayed care and treatment. She was brought from the DRC to the health center in Uganda after her family found she had already been ailing. Pneumonic plague in adults is usually attributed to delayed diagnosis of bubonic plague and subsequent disease progression; the average time from full onset of pneumonic plague to death is 1.9 days [
19]. Because of the rapid progression to death, it is essential to obtain laboratory diagnosis and treatment as quickly as possible [
21]. However, the nonspecific presentation of pneumonic plague can complicate diagnosis [
19], and having access to field-appropriate diagnostics in low-resource settings is critical to preventing plague deaths. Plague RDT is an field-appropriate tool that has led to early detection of outbreaks and rapid implementation of control measures in multiple countries in Africa [
4].
Historical documentation of plague in Africa dates back to the early 1900s [
22], though it likely occurred well before then. These outbreaks have ranged in size from dozens to thousands of cases. A 2017 outbreak in Madagascar ultimately affected over 2,000 persons, with 202 deaths (case-fatality rate=8.6%)[
23]; this outbreak spread quickly in part due to a weak surveillance system [
11]. In contrast, the outbreak in Zombo District was likely contained by early detection and response in the form of isolation and treatment of the suspected case, contact tracing, and prophylaxis for contacts. Having access to tools for early detection and treatment, as well as ensuring adequate training of health workers, are critical to addressing plague outbreaks and reducing overall plague deaths [
19]. The Ministry of Health should focus efforts on sustained surveillance for early detection and timely response to outbreaks of this nature.
Conclusion


This outbreak of plague affected one family in Uganda and was imported from the DRC. Timely response in form of prompt identification, isolation, and treatment of case-persons, tracing of contacts, and antibiotic prophylaxis for the contacts were effective in containing this outbreak.
Recommendations
We recommended that the Ministry of health continues to foster sustainable surveillance efforts to enable prompt detection; putting in place early warning systems including continuous community sensitization and provision of more RDT kits to major health facilities in the West Nile region. Because the outbreak was imported, strengthening of cross-border collaborations and surveillance led by the Ministries of Health in both Uganda and DRC to ensure early detection and containment of plague outbreaks is equally important. This recommendation is strengthened by evidence of gains realised during the Ebola Virus Disease outbreak in DRC during 2018-2019
What is known about this topic
- Plague is a zoonotic disease that dates way back. Previous outbreaks have proved how dangerous a disease it is, existing in different forms and mode of transmission making it fatal.
What this study adds
- The need for collaborative efforts using a One Health approach including cross border collaborations for enhanced surveillance and early detection of outbreaks and therefore quick response to avert widespread outbreaks, morbidity and mortality.
Competing interests


The authors declare no competing interests.
Authors' contributions


DNG took lead in execution of the investigation. She wrote the drafts of the manuscript and revised the paper for substantial intellectual content. BMB and SN participated in the conceptual design and development of the investigation and were involved in field data collection as well as data analysis. They also reviewed the paper for substantial intellectual content. BK, LB, ARA, participated in designing the investigation, supervision of field data collection, data analysis, and reviewed the draft manuscript for substantial intellectual content. All authors have read and approved the final manuscript.
Acknowledgments


We acknowledge the Zombo District Local Government for their technical support and effort during the response to this outbreak. We appreciate the Uganda Virus Research Institute for specimen testing to confirm the etiology of this outbreak.
Figures


Figure 1: Map showing movement of case-patient A from Mahagi in DRC to Warr HCIII in Zombo, Uganda. Four plague cases were reported in Mahagi, Ituri Province, one of whom traveled to Warr HCII for treatment on 28th March 2019
References


- CDC. Plague home. CDC. 2019. Accessed May 2020.
- CDC. Plague home. CDC. 2021. Accessed May 2021.
- WHO. Plague. WHO. Accessed May 2020.
- Forrester JD, Apangu T, Griffith K, Acayo S, Yockey B, Kaggwa J, Kugeler KJ, Schriefer M, Sexton C, Ben Beard C, Candini G, Abaru J, Candia B, Okoth JF, Apio H, Nolex L, Ezama G, Okello R, Atiku L, Mpanga J, Mead PS. Patterns of Human Plague in Uganda, 2008-2016. Emerg Infect Dis. 2017 Sep; 23(9):1517-1521. https://dx.doi.org/10.3201/eid2309.170789
- American Public Health Association (APHA). Control of Communicable Diseases Manual. APHA. Accessed Mar 2019.
- Mensah P, Mwamakamba L, Kariuki S, Fonkoua MC, Aidara-Kane A. Strengthening foodborne diseases surveillance in the WHO African region: An essential need for disease control and food safety assurance. African Journal of Food, Agriculture, Nutrition and Development. 2012; 12(4):6336-53. https://doi.org/10.18697/ajfand.52.WHO-7
- Kenneth L. Gage. Plague and Other Yersinia Infections. Editor(s): Lee Goldman, Andrew I. Schafer.Goldman's Cecil Medicine. 24th ed. Philadelphia: W.B. Saunders; 2012. 1895-1900 p.https://doi.org/10.1016/B978-1-4377-1604-00320-1 .
- Munyenyiwa A, Zimba M, Nhiwatiwa T, Barson M. Plague in Zimbabwe from 1974 to 2018: A review article. PLoS Negl Trop Dis. 2019 Nov 21; 13(11):e0007761.https://doi.org/10.1371/journal.pntd.0007761 PubMed | Google Scholar
- DENNIS DT, MEAD PS. Plague. Tropical Infectious Diseases. 2006: 471-81.https://dx.doi.org/10.1016/B978-0-443-06668-9.50047-8 PubMed | Google Scholar
- Eisen RJ, MacMillan K, Atiku LA, Mpanga JT, Zielinski-Gutierrez E, Graham CB, Boegler KA, Enscore RE, Gage KL. Identification of risk factors for plague in the West Nile Region of Uganda. Am J Trop Med Hyg. 2014 Jun;90(6):1047-58.https://dx.doi.org/10.4269/ajtmh.14-0035 PubMed | Google Scholar
- Drancourt M, Raoult D. Investigation of Pneumonic Plague, Madagascar. Emerg Infect Dis. 2018 Jan;24(1):183. https://dx.doi.org/10.3201/eid2401.170760 PubMed | Google Scholar
- Abedi AA, Shako JC, Gaudart J, Sudre B, Ilunga BK, Shamamba SKB, Diatta G, Davoust B, Tamfum JM, Piarroux R, Piarroux M. Ecologic Features of Plague Outbreak Areas, Democratic Republic of the Congo, 2004-2014. Emerg Infect Dis. 2018 Feb;24(2):210-220.https://dx.doi.org/10.3201/eid2402.160122 PubMed | Google Scholar
- Uganda Ministry of Health. National Technical Guidelines for Intergrated Disease Surveillance and Response, Third Edition. Uganda, MOH. 2021.
- Gov't of Uganda, UNHCR. Uganda Refugee Response Plan (RRP) 2019-2020. Relief Web. 2019. Accessed Nov 2020.
- WHO. Implementation and management of contact tracing for Ebola virus disease: emergency guideline. WHO. 20 Accessed Nov 2020.
- News Desk. Plague: A dozen additional cases reported in Ituri province, DRC. Outbreak News Today. 2019. Accessed May 2020.
- News Desk. Plague cluster reported in Ituri, Democratic Republic of Congo. Outbreak News Today. 2020. Accessed Jul 2020.
- Forrester JD, Apangu T, Griffith K, Acayo S, Yockey B, Kaggwa J, Kugeler KJ, Schriefer M, Sexton C, Ben Beard C, Candini G, Abaru J, Candia B, Okoth JF, Apio H, Nolex L, Ezama G, Okello R, Atiku L, Mpanga J, Mead PS. Patterns of Human Plague in Uganda, 2008-2016. Emerg Infect Dis. 2017 Sep;23(9):1517-1521. https://dx.doi.org/10.3201/eid2309.170789 PubMed | Google Scholar
- Andrianaivoarimanana V, Piola P, Wagner DM, Rakotomanana F, Maheriniaina V, Andrianalimanana S, Chanteau S, Rahalison L, Ratsitorahina M, Rajerison M. Trends of Human Plague, Madagascar, 1998-2016. Emerg Infect Dis. 2019 Feb;25(2):220-228.https://dx.doi.org/10.3201/eid2502.171974 PubMed | Google Scholar
- WHO. Early detection, assessment and response to acute public health events: Implementation of Early Warning and Response with a focus on Event-Based Surveillance. WHO. 2014. Accessed Dec 2021. Google Scholar
- WHO. Revamp of the plague detection in Madagascar yields quick and sustainable wins. WHO. Accessed Apr 2019.
- DAVIS DH. Plague in Africa from 1935 to 1949; a survey of wild rodents in African territories. Bull World Health Organ. 1953; 9(5):665-700. PubMed | Google Scholar
- WHO. Plague - Madagascar. WHO. 2017. Accessed Jun 2020.