Epidemiology of Dysentery, Uganda, 2014-2018
Maureen Nabatanzi1,&, Benon Kwesiga1, Lilian Bulage1, Bernard Lubwama2, Alex Riolexus Ario1, Julie Harris3
1Uganda Public Health Fellowship Program, Ministry of Health, Kampala, Uganda, 2Integrated Epidemiology, Surveillance and Public Health Emergencies Department, Ministry of Health, Kampala, Uganda, 3US Centers for Disease Control and Prevention, Kampala, Uganda
&Corresponding author
Maureen Nabatanzi, Uganda Public Health Fellowship Program, Ministry of Health, Kampala, Uganda. mnabatanzi@musph.ac.ug
Introduction:
Shigella dysenteriae infection (dysentery) is endemic in developing countries. In Uganda, dysentery is a priority disease for surveillance, however, there is a paucity of data on its magnitude and distribution. We used surveillance data to describe dysentery cases in Uganda, 2014-2018.
Methods:
We extracted dysentery surveillance data for 2014-2018 from the national Health Management Information System (HMIS) database. The HMIS defines suspected dysentery as diarrhea with visible blood. Confirmed dysentery is a suspected case with Shigella dysenteriae type 1 cultured from stool. We evaluated incidence by age, sex, and district over the study period.
Results:
Between 2014-2018, Uganda reported 415,337 suspected dysentery cases, of which 52,549 (13%) were laboratory confirmed. From 2014 to 2018, the annual incidence of suspected cases declined from 29/10,000 to 16/10,000 (p=0.001), while the proportion of cases with laboratory confirmation increased from 6.1% to 29%. Across all years, children under five years were most affected, and females were more affected than males. Confirmed Shigella dysentery coincided with rainy seasons, with annual surges during the rainy seasons of March, July, and late September-December. Karamoja sub-region in Northern Uganda consistently reported the highest annual incidence of suspected dysentery (annual incidence 41 to 219/10,000).
Conclusion:
During 2014-2018, annual incidence of suspected dysentery in Uganda declined while proportion of cases confirmed increased; disease peaks occurred during rainy seasons. We recommend that Ministry of Health investigates the specific risk factors for dysentery during rainy seasons to inform prevention measures and identifies approaches to improve laboratory confirmation capacity.
Introduction

Dysentery is a syndrome characterized by diarrhea with visible blood and mucous. It is endemic in developing countries and overcrowded settings with unsafe water and poor sanitation [1, 2]. Among the very young and the elderly, the case-fatality rate may approach 20%, primarily due to severe dehydration [2]. According to a global foodborne disease burden report by the World Health Organization (WHO), more than 51 million cases of dysentery were reported in 2010. In the WHO Africa Sub-region (East), in which Uganda lies, dysentery contributes to 13% of all foodborne diseases [3].
Shigella dysenteriae is an acute bacterial disease that is the most common cause of dysentery. It is characterized by diarrhea accompanied by fever, nausea, vomiting, and cramps, referred to as shigellosis. Blood and mucous in the diarrhea are a result of microorganisms´ invasion and destruction of cells lining the small intestine and colon [
4]. However, dysentery can be caused by many different organisms beyond
Shigella spp., including enterohemorrhagic
Escherichia coli serotype O157:H7,
Campylobacter jejuni, enteroinvasive
E. coli, Salmonella spp., and
Entamoeba histolytica [
5].
Shigella spp. is divided into four serogroups with multiple serotypes: A (
S. dysenteriae, 15 serotypes, the primary cause of dysentery); and B (
S. flexneri, 8 serotypes), C (
S. boydii, 19 serotypes), and D (
S. sonnei, 1 serotype), all of which cause dysentery much less frequently than
S. dysenteriae serotype 1 [
1,
4].
Shigella dysenteriae serotype 1 (Group A) is transmitted through the fecal-oral route and produces Shigatoxin type 1, which is believed to be responsible for the severity of symptoms associated with infection [
4]. Infected persons develop disease in 1-3 days after infection, with mild and asymptomatic infections being self-limiting and lasting an average of four to seven days. Symptomatic or asymptomatic persons may spread infection to others through direct fecal oral-transmission, or when they contaminate food, water, fomites (utensils, toys, and clothes) after failing to wash hands thoroughly after defecation [
6,
7].
Due to its epidemic potential, Integrated Disease Surveillance and Response (IDSR) guidelines list dysentery as a priority disease for surveillance [
8]. However, the trends and distribution of dysentery among sub-population groups and regions in Uganda are not well documented. We described dysentery cases in Uganda from 2014 to 2018 using surveillance data, with the objectives of informing planning, control, and prevention measures.
Methods


Study setting
Uganda is an East African country of approximately 44 million persons [
9]. Uganda is divided into four administrative regions and 11 sub-regions as follows: Northern region (Karamoja, West Nile, Acholi, and Lango), Western region (Western and South Western), Eastern region (Teso, Elgon and East Central), and Central region (Central 1 and Central 2). The sub-regions are further divided into districts. Uganda was divided into 112 districts in 2014, 128 districts in 2015, and 135 districts in 2019. According to Uganda´s 2016 National Household Survey, 22% of households lacked access to improved water sources (those that are protected from outside contamination, such as piped water); while 7% of households practiced open defecation [
10].
Study design
We analyzed routinely-collected surveillance data reported through the Health Management Information System (HMIS) of the MoH from 2014 to 2018.
The dysentery surveillance system
According to the HMIS, a suspected case is a person with diarrhea with visible blood in stool. A confirmed case is a suspected case with stool culture-positive for
Shigella dysenteriae type 1. At all health facility outpatient and inpatient departments, standardized HMIS data collection tools are used to record suspected and confirmed cases. Weekly data on suspected dysentery cases are recorded in the health unit weekly epidemiological form, ‘HMIS 033b’;. Monthly data on laboratory confirmed
Shigella dysentery cases are recorded in the health unit outpatient monthly report form, ‘HMIS 105’. Health workers send these data to their district biostatisticians, who then upload it into the national electronic HMIS database, called the District Health Information System version 2 (DHIS2) [
8]. The proportion of health facilities that report suspected dysentery weekly, monthly and annually is tracked by DHIS2 and computed as district and national reporting rates for HMIS 033b. In DHIS2, reporting rate is computed as the number of health facilities in Uganda that reported on suspected dysentery in a given period out of the total number of health facilities in Uganda expected to report on suspected dysentery in the same period of time.
Data abstraction
In DHIS2, we downloaded ‘HMIS 033b’ weekly suspected dysentery case counts organized by district, age-group and sex for the years 2014 to 2018. Similarly, in DHIS2, we downloaded ‘HMIS 105’ monthly confirmed
Shigella dysentery case counts for the years 2014 to 2018. We also downloaded annual reporting rates for the ‘HMIS 033b’ data from DHIS2 to describe the reporting trends and compare with the estimated incidence over time.
Data analysis
We analyzed data using Microsoft Excel 2019
™ and EpiInfo
™ software version 7.2. We conducted descriptive analysis of the dysentery cases by person, place, and time. Age was categorized into 0 to 4 years, 5 to 59 years, and 60 years and above. As reporting of age data using these categories did not begin until 2016, analysis of suspected dysentery by age group in 2014 and 2015 was omitted from the analysis. We used the age, sex, and district-specific population census data as the denominators to calculate specific incidence. Population data were extracted from the 2014 National Population and Housing Census which used a 3% annual growth rate to calculate projections [
9]. We plotted trends in incidence of suspected dysentery, confirmed
Shigella dysentery, and national reporting rate and used logistic regression to test the significance of observed trends at p<0.05. We compared occurrence of suspected dysentery to Uganda´s two rainy seasons, which occur during March-May and September-December [
11]. We used Quantum Geographic Information System (QGIS
™) software version 3.4.8 to present the annual incidence of suspected dysentery, and the proportion of suspected dysentery that was confirmed as
Shigella on maps.
Availability of supporting data
The datasets upon which our findings are based belong to the Uganda Public Health Fellowship Program. For confidentiality reasons the datasets are not publicly available. However, the data sets can be availed upon reasonable request from the corresponding author and with permission from the Uganda Public Health Fellowship Program.
Ethical consideration
This analysis used aggregated routine surveillance data reported by health facilities for the Uganda MoH and were used with permission of the MoH. The Office of the Associate Director for Science, Centers for Global Health, CDC, cleared this activity as not human subject research, and its primary intent was public health practice or a disease control activity. Accordingly, the CDC provided a Non-Research Determination under Project Identification number 0900f3eb819c0e4d. For confidentiality and privacy, access to the MoH data bank (DHIS2) requires a password. The surveillance data extracted from DHIS2 were aggregated with no individual patient identifiers and stored on a password protected computer only accessible to the study team. Since completion of the analysis in 2020, data were stored for three years on a password protected computer before destruction for security purposes.
Results


Incidence of dysentery, Uganda, 2014-2018
A total of 415,337 suspected cases of dysentery (54,549 confirmed
Shigella; 13%) were reported from January 2014 to December 2018. Annual incidence of suspected dysentery declined from 29/10,000 in 2014 to 16/10,000 in 2018 (p=0.001). Over the same period, there was an increase in annual confirmed
Shigella dysentery cases, from 1.8/10,000 to 4.6/10,000 (p=0.001)
Table 1.
There was a steady rise in annual national reporting of suspected dysentery from 52% in 2014 to 81% in 2018
Figure 1. We observed annual surges in March, July and late September-October
Figure 2. The surges coincided with Uganda´s two rainy seasons in March-May and September - December. From 2016 to 2018, children under five consistently had the highest incidence of suspected dysentery (ranging from 32-38/10,000), while persons aged 5-59 years consistently had the lowest incidence (ranging from 12-17/10,000)
Figure 3. Males consistently had lower annual incidence of suspected dysentery than females, although incidence decreased across both sexes
Figure 4.
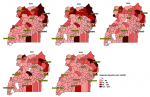
The decline over time is clearly visible in the annual maps of Uganda
Figure 5. Northern Uganda´s sub-region of Karamoja was consistently the most affected subregion, with multiple districts reporting annual incidence of suspected dysentery well above 40 per 10,000 people. The Western and South Western sub-region districts were less affected, typically having incidence of 40 per 10,000 or lower. In 2014 and 2015, some districts in Karamoja had incidence up to 219 per 10,000, and this region continued to have high incidence of suspected dysentery throughout the evaluation period. By 2018, nearly all districts in the country were reporting incidence of 40 per 10,000 or lower, except those in Karamoja.
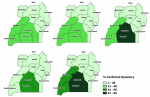
Proportion and distribution of laboratory confirmed Shigella dysentery cases by sub-region and year Uganda, 2014 to 2018
Nationally, confirmed cases increased as a proportion of the total (suspected) cases, from 6.1% in 2014 to 29% in 2018
Table 1. The Northern sub-regions (West Nile, Acholi, Karamoja and Lango) consistently reported the lowest proportion of suspected cases with laboratory confirmation (<20%)
Figure 6. Western and South Western sub-regions demonstrated increasing proportions of suspected cases with laboratory confirmation, rising from 22% in 2014 to 45% in 2018. Sub-regions Central 1 and 2 consistently reported the highest percentage of suspected cases that were laboratory-confirmed, rising from 26% in 2014 to 65% in 2018
Figure 6.
Discussion


From 2014-2018, the annual incidence of suspected dysentery declined significantly across Uganda, while the proportion of cases that were laboratory-confirmed Shigella increased. Females and young children were most affected. Specific districts, especially those in the north of Uganda, still appear to struggle with high levels of dysentery and low rates of laboratory confirmation.
Dysentery occurrence was seasonal, with surges that coincided with Uganda´s rainy seasons. Rainfall, in addition to temperature and relative humidity, can be a predictor for dysentery and other diarrheal diseases, particularly when there is flooding [
12]. When populations use unimproved water sources such as rivers, ponds, or open wells, flooding may allow the entry of infected feces, increasing the risk of dysentery [
13,
14]. In 2016, approximately one quarter of Uganda´s households used unimproved water sources [
10]. Beyond the provision of improved water sources, encouraging actions such as chlorinating and boiling water for drinking and proper use of toilets are known to reduce the impact of diarrheal diseases [
15]. Enhancing the use of these approaches in Uganda could reduce the impact of a variety of diarrheal diseases.
Northern regions in general, and particularly the Karamoja sub-region, had both the highest rates of dysentery and the lowest rates of laboratory confirmation of suspected cases. The Karamoja sub-region is among the poorest in Uganda, and has suffered from increasing poverty in recent years even as national poverty levels have declined [
16]. Indeed, the population in this area suffers not only from diarrheal diseases, but also food insecurity and malnutrition [
17,
18]. Despite an increase in availability of safe water in recent years in this region [
10], sanitation is still extremely poor, with two-thirds of households practicing open defecation [
10]. Because of the high rates of diarrheal diseases in this area, laboratory confirmation may be particularly important to identify the causative agents and design appropriate treatment and preventive interventions.
Failure to obtain laboratory confirmation can lead to misclassification and ineffective treatments [
15]. Furthermore, laboratory confirmation of cases is necessary to establish individual antimicrobial susceptibility profiles, guide treatment regimens [
15,
19], and determine population antimicrobial resistance patterns [
20]. Although Uganda established the National Health Laboratory Services (NHLS) to coordinate improvement of laboratory capacity; as of 2010, most laboratories did not meet national requirements for equipment, supplies, and skilled human resources [
19]. Northern Uganda and specifically Karamoja sub-region has struggled with civil disputes, remoteness, and poor infrastructure that have adversely affected health service delivery [
21]. There is a need to identify approaches to improve laboratory capacity and services in light of these limitations, especially in Northern Uganda.
While other diarrheal diseases - notably, cholera [
22-26] - receive substantial attention in Uganda, to our knowledge there have been no published analyses on the epidemiology of dysentery in Uganda to date. The spotlight on cholera has led to a national plan for cholera control [
27] and investigations to identify hotspot areas and multiple subsequent interventions, including Oral Cholera Vaccination (OCV) campaigns [
25,
27]. Indeed, despite a much higher burden of dysentery than of cholera [
25], no specific guidelines exist for dysentery prevention in Uganda. Using findings from this analysis, the Ministry of Health should intensify efforts to track, investigate, and report on dysentery in Uganda, and can call attention and prioritize resources for this under-recognized issue.
Limitations
This study relied on surveillance data routinely reported from health facility level to national level, which may have had reporting errors. However, MoH conducts weekly verification exercises for the ‘HMIS 033b’ data to reduce erroneous reports in the system. Descriptive analysis for age was limited to three broad age categories provided on the monthly outpatient form, ‘HMIS 105’ during 2016 - 2018. In 2020, the MoH revised HMIS 105 to include more specific age groups. Future studies should consider descriptive analysis of dysentery using specific age categories to inform age appropriate interventions. The suspect case definition for dysentery used included all persons presenting with bloody diarrhea while the confirmed case definition was stool culture-positive for
Shigella dysenteriae type 1. This could have introduced misclassification bias since bloody diarrhea is also a symptom of dysentery caused by other
Shigella dysenteriae types and non-bacterial organisms. Prospective studies on burden of dysentery in Uganda should include other bloody diarrhea causing organisms. Although population-based data studies may be liable to misclassification, this analysis describes dysentery occurrence; knowledge of which was inadequate in Uganda.
Conclusion


Although suspected dysentery in Uganda has declined, females, children under five and the Karamoja sub-region continue to be highly affected. In addition, the proportion of suspected dysentery cases that are laboratory confirmed remains low in Uganda. Occurrence of dysentery tends to be seasonal, coinciding with Uganda's rainy seasons. Risk factors for dysentery in Uganda especially during the rainy seasons should be further investigated, and the capacity to conduct laboratory confirmation improved.
What is known about this topic
- Dysentery is a disease of public health concern in developing countries with poor water and sanitation
- Epidemiology of dysentery in Uganda has not been well described
What this study adds
- A description of the trend in suspected dysentery in Uganda during 2014-2018 using surveillance data that reveals seasonality and higher burden among females, children under five and the Karamoja sub-region
- The low proportion of suspected dysentery cases that are laboratory confirmed in Uganda necessitates interventions to improve diagnosis of dysentery
Competing interests


The authors declare no competing interests.
Funding and disclaimer
This work was funded by the Cooperative Agreement-Provision of Comprehensive HIV/AIDS services and Developing National Capacity to manage HIV/AIDS Programs in the Republic of Uganda under the President's Emergency Plan for AIDS Relief (Cooperative Agreement number U2GGH001353–04) through the United States Centers for Disease Control and Prevention to Uganda Ministry of Health through Makerere University School of Public Health. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the US Centers for Disease Control and Prevention, the Department of Health and Human Services, Makerere University School of Public Health, or the MoH. The staff of the funding body provided technical guidance in the design of the study, ethical clearance and collection, analysis, and interpretation of data and in writing the manuscript.
Authors' contributions


MN conceived and designed the study, acquired, analyzed, interpreted the data and wrote the first draft of the manuscript. BL contributed substantially to analysis and interpretation of data. BK, LB, ARA and JH critically reviewed the paper for important intellectual content. All authors read and approved the manuscript.
Acknowledgments


We would like to acknowledge the responsible departments of the MoH for allowing us to use the routinely generated data.
Tables and figures


Table 1: Suspected and laboratory-confirmed Shigella dysentery cases in Uganda, 2014 - 2018
Figure 1: Trends in national incidence and reporting rate of suspected dysentery in Uganda, 2014 - 2018
Figure 2: Suspected dysentery cases by month in Uganda, 2014-2018
Figure 3: Trend of incidence of suspected dysentery by age group, Uganda, 2016 to 2018
Figure 4: Trend of incidence of suspected dysentery by sex in Uganda, 2014 to 2018
Figure 5: Trend of incidence of suspected dysentery by district in Uganda, 2014 to 2018
Figure 6: Percentage of suspected dysentery cases that were laboratory-confirmed Shigella, by sub-region in Uganda, 2014-2018
References


- Hale TL, Keusch GT. Shigella. In: Baron S, editor. Medical Microbiology. 4th edition. Galveston (TX): University of Texas Medical Branch at Galveston; 1996. Chapter 22. PubMed | Google Scholar
- Ministry of Health, Uganda. National Technical Guidelines for Integrated Disease Surveillance and Response. Uganda: Ministry of Health. 2012 [cited 2022 Nov 15].
- WHO. WHO Estimates of the Global Burden of Foodborne Diseases: Foodborne Disease Burden Epidemiology Reference Group 2007-2015. World Health Organization. 2015 [cited 2022 Nov 15].
- American Public Health Association (APHA). Control of Communicable Diseases Manual: Shigellosis. 20 ed. Washington DC: American Public Health Association; 2015.
- CDC. Laboratory Methods for the Diagnosis of Epidemic Dysentery and Cholera. Atlanta, Georgia CDC. 1999 [cited 2022 Nov 15].
-
Islam MS, Hossain MA, Khan SI, Khan MN, Sack RB, Albert MJ, Huq A, Colwell RR. Survival of Shigella dysenteriae type 1 on fomites. J Health Popul Nutr. 2001 Sep;19(3):177–82. Google Scholar
- CDC. CDC Yellow Book 2020: Health Information for International Travel. Mark D. Gershman JES, editor. USA: Centers for Disease Control and Prevention; 2019.
- Ministry of Health. The Health Management Information System. Kampala, Uganda: MOH Resource Centre. 2014 [cited 2022 Nov 15].
- Uganda Bureau of Statistics (UBOS). National Population and Housing Census 2014. UBOS. 2014 [cited 2022 Nov 15].
- UBOS. Uganda National Household Survey 2016/17. UBOS. 2018 [cited 2022 Nov 15].
- World Bank Group. Climate Change Knowledge Portal: Country - Uganda, Current Climate Climatology. World Bank Group. 2021 [cited 2022 Nov 15].
- Wu X, Liu J, Li C, Yin J. Impact of climate change on dysentery: Scientific evidences, uncertainty, modeling and projections. Science of The Total Environment. 2020[cited 2022 Nov 15]; 714:136702. https://doi.org/10.1016/j.scitotenv.2020.136702. Google Scholar
- Cabral JP. Water microbiology. Bacterial pathogens and water. Int J Environ Res Public Health. 2010 Oct[cited 2022 Nov 15]; 7(10):3657-703.https://doi.org/10.3390/ijerph7103657 PubMed | Google Scholar
- Ni W, Ding G, Li Y, Li H, Liu Q, Jiang B. Effects of the floods on dysentery in north central region of Henan Province, China from 2004 to 2009. Journal of Infection. 2014[cited 2022 Nov 15]; 69(5):430-9. https://doi.org/10.1016/j.jinf.2005.016 . Google Scholar
- World Health Organization. Guidelines for the control of shigellosis, including epidemics due to Shigella dysenteriae type 1. World Health Organization. 2005 [cited 2022 Nov 15].
.
- World Bank. The Uganda Poverty Assessment Report 2016. World Bank. 2016 [cited 2022 Nov 15].
.
- Food and Agriculture Organization (FAO). Responding to the Challenges of Poverty, Food Insecurity and Climate Change: Country Programming Framework 2015-2019. FAO. 2015 [cited 2022 Nov 15].
.
- Kirabira P, Okeyo D, Ssempebwa J. Risk Factors to Persistent Dysentery among Children under the Age of Five in Rural Sub-Saharan Africa; the Case of Kumi, Eastern Uganda. Food Science & Nutrition Research. 2018[cited 2022 Nov 15]; 1:1-6. http://dx.doi.org/10.33425/2641-4295.1003 Google Scholar
- Ministry of Health, Uganda. Uganda National Health Laboratory Services Strategic Plan (2010-2015). Kampala, Uganda: Ministry of Health. 2010. [cited 2022 Nov 15].
.
- Hegde S, Benoit SR, Arvelo W, Lindblade K, López B, McCracken JP, Bernart C, Roldan A, Bryan JP. Burden of laboratory-confirmed shigellosis infections in Guatemala 2007-2012: results from a population-based surveillance system. BMC Public Health. 2019[cited 2022 Nov 15]; 19(3):474. https://doi.org/10.1186/s12889-019-6780-7 PubMed | Google Scholar
- United Nations Population Fund (UNFPA). Population Matters: Leaving no one behind in Karamoja. United Nations Population Fund. 2018 [cited 2022 Nov 15].
.
- Legros D, McCormick M, Mugero C, Skinnider M, Bek'Obita DD, Okware SI. Epidemiology of cholera outbreak in Kampala, Uganda. East African Medical Journal. 2000[cited 2022 Nov 15]; 77(7):347-9. https://doi.org/10.4314/eamj.v77i7.46659 Google Scholar
- Iramiot JS, Rwego IB, Kansiime C, Asiimwe BB. Epidemiology and antibiotic susceptibility of Vibrio cholerae associated with the 2017 outbreak in Kasese district, Uganda. BMC Public Health. 2019 Oct 29[cited 2022 Nov 15]; 19(1):1405. https://doi.org/10.1186/s12889-019-7798-6 PubMed | Google Scholar
- Kwesiga B, Pande G, Ario AR, Tumwesigye NM, Matovu JKB, Zhu BP. A prolonged, community-wide cholera outbreak associated with drinking water contaminated by sewage in Kasese District, western Uganda. BMC Public Health. 2017 Jul 18[cited 2022 Nov 15]; 18(1):30. https://doi.org/10.1186/s12889-017-4589-9 PubMed | Google Scholar
-
Bwire G, Ali M, Sack DA, Nakinsige A, Naigaga M, Debes AK, Ngwa MC, Brooks WA, Garimoi Orach C. Identifying cholera "hotspots" in Uganda: An analysis of cholera surveillance data from 2011 to 2016. PLoS Negl Trop Dis. 2017[cited 2022 Nov 15]; 11(12):e0006118. https://doi.org/10.1371/journal.pntd.0006118 PubMed | Google Scholar
- Ministry of Health, Uganda. Cholera Prevention and Control Strategic Plan 2017/18. Kampala, Uganda: Ministry Of Health. 2017 [cited 2022 Nov 15].
.
- WHO. Uganda targets over 1,600,000 persons for Cholera vaccination campaign. World Health Organization. 2018 [cited 2022 Nov 15].