Seroprevalence of leptospirosis among presumptive malaria patients in a secondary health facility in Oyo state, Southwest Nigeria
Mathias Besong1,&, Emmanuel Awosanya2, Olusoji Adeyanju3, Adedayo Adigun3, Charles Michael4, Muhammed Balogun4, Gabriel Ogundipe2
1Nigeria Field Epidemiology and Laboratory Training Programme, Abuja, Nigeria, 2University of Ibadan, Ibadan, Oyo State, Nigeria, 3Adeoyo Maternity Teaching Hospital Yemetu, Ibadan, Oyo State, Nigeria, 4African Field Epidemiology Network, Abuja, Nigeria
&Corresponding author
Mathias Besong, Nigeria Field Epidemiology and Laboratory Training Programme, Abuja, Nigeria. drbisong@yahoo.com
Introduction:
Leptospirosis is a neglected tropical zoonoses that presents with fever and can be misdiagnosed, with fatal outcomes. Its incidence has been on the increase in recent times with 1 million cases and over 60,000 deaths reported annually worldwide. We determined the seroprevalence of leptospirosis and associated risk factors among presumptive malaria patients in a secondary health facility in Ibadan.
Methods:
We used Leptospira Rapid Diagnostic Test kit to estimate the seroprevalence of leptospirosis among 143 patients who presented at the study center between 27th April and 15th May 2019 whom a physician had seen and presumed they had malaria. We used one drop of blood for the test from blood collected by trained laboratory technicians for malaria diagnosis. Data on patients' demographics and risk factors were collected using an interviewer-administered questionnaire. We calculated frequencies, means and proportion and also calculated odds ratios at 95% confidence interval (α=0.05).
Results:
The median age of the participants was 34 years (range: 6 months-80 years). Eighty-nine (62.2%) of them were females. Of the 143 tested, 12 (8.4%) were positive for Leptospira IgM/IgG antibodies. Eleven (7.7%) patients were positive for malaria. One (0.7%) was positive for both malaria and leptospirosis. Owning a backyard garden was significantly associated with leptospirosis (OR: 3.7, CI: 1.01-12.92).
Conclusion:
We confirmed Leptospirosis among febrile patients at the study center We found that the seroprevalence of leptospirosis is same as that of malaria among the sampled population. However, leptospirosis/malaria co-infection was low. We recommend a review of the diagnostic protocol to include leptospirosis as a differential.
Introduction

Leptospirosis is one of the most important and most widespread emerging or re-emerging zoonotic diseases that has a considerable impact on human health with multiple outbreaks reported in all continents [1, 2]. About 1 million cases of the diseases are reported annually with over 60,000 deaths, most of which occur in tropical and sub-tropical climates [1]. However, the number of reported cases have been shown to be increasing globally. In Malaysia, for example, in 2004 they reported only 248 cases of leptospirosis compared to over 3,600 in 2012. This increase was because of changes in surveillance and diagnostic practices after introducing leptospirosis as a notifiable disease in 2010 [3]. Thus supporting the fact that there is a possibility of gross under reporting of the disease, especially in resource-limited settings where there is usually poor awareness of the disease, and lack of adequate diagnostic facilities [4, 5].
Spread of Leptospira organism to humans occur mainly by direct contact with an infected animal or indirectly through contact with contaminated soil or water through mucous membranes or broken skin [
6,
7]. Researchers relate spread of the disease to increased rainfall, livestock holding, increase rodent activities, poor hygiene practices, inadequate refuse disposal practices and overcrowding, conditions characteristic of urban slums in the developing world [
8,
9].
Leptospirosis is endemic in both wild and domestic animals in Africa. It is a major cause of febrile illness with about 750,000 new cases reported annually from various parts of the continent. However, most of them directly result from high urban growth rates and indiscriminate spread of shantytowns, especially around the flood-prone areas along the Atlantic coast [
9]. In sub-Saharan Africa, the increasing prevalence is because of a combination of climate change, increasing risk of flooding, population growth, and urbanization5. The prevalence of leptospirosis among febrile patients in Nigeria is poorly documented [
9]. However, previous studies in “healthy” humans in some parts of the country puts it at between 13.5 and 20.4% [
10,
11].
The common clinical signs/symptoms of the disease are fever, headache, myalgia, conjunctivitis, nausea, vomiting, diarrhea, abdominal pain, cough and sometimes skin rashes [
12,
1]. The disease even in mild forms can cause foetal complications, including foetal deaths or abortion [
13,
14]. The above signs and symptoms mimic many common diseases (malaria, typhoid fever, hepatitis B, cholera) but malaria is the usual presumptive diagnosis among febrile patients in many African settings [
15]. There is therefore, a high probability of misdiagnosis and under-diagnosis of leptospirosis, sometimes with fatal outcomes. The aim of this study was, therefore, to determine the seroprevalence of leptospirosis among presumed malaria patients at a secondary health facility in Ibadan, Oyo State, Southwest, Nigeria
Methods


Study area
We conducted the study among febrile patients seeking medical care at the Out-Patients Department (OPD) of Adeoyo Maternity Teaching Hospital (AMTH), Yemetu, Oyo State between 27th April and 15th May 2019, whom a physician had seen and had made a presumptive diagnosis of malaria. AMTH is located in Ibadan North Local Government Area (LGA). The health facility is surrounded by classical urban slums, characterized by high rodent activities, poor hygiene practices, inadequate refuse disposal practices and overcrowding. The surrounding areas are also prone to flooding from blockage of drainages and the activities of the great Ogunpa river. The hospital receives patients from all social classes but mainly people of lower income level because of the highly subsidized treatment fees from the state government.
Study design
The study is a cross-sectional study with patients recruited consecutively. We included all patients with history of fever that has lasted at least 24-48 hours with or without headache, malaise, abdominal pain, vomiting, jaundice, whom a clinician has examined and referred to the laboratory for malaria test but had taken no prior medication for the current illness. We excluded those without fever and those with fever but were either on conventional treatment or were not referred to the laboratory for malaria test by any of the consulting physicians on duty.
Sample size calculation
We recruited 143 participants for this study. This was based on the fact that we calculated a minimum sample size of 139 using the formula for estimation of proportions at a prevalence of 10% [
9,
16].
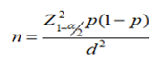
Where;
P = Prevalence of leptospirosis among febrile patients
Z∝ = Standard normal value corresponding to 95% confidence level (set at 1.96).
d = Sampling error (set at 5%)
Sample collection
Using a transparent rubber dropper, one drop of blood was collected from the blood already collected by trained a laboratory technician and stored in a properly labelled EDTA bottle. The blood was dropped into the designated portion of leptospirosis RDT kits placed parallel on the laboratory bench. The Leptospira IgM/IgG used was manufactured by AllTest Co
®. China with a relative sensitivity of 93.8% (95% CI*: 69.8% - 99.8%) and relative specificity of 98.7% (95% CI*: 96.1% - 99.7%). Accuracy: 98.3% (95% CI*:95.8% - 99.5%). Results for leptospirosis was recorded as positive IgG, positive IgM, positive IgG/IgM and negative. With a separate rubber dropper, another drop of blood from the same sample bottle was applied on the Malaria RDT kit (SD BIOLINE) also placed parallel on the bench. This was done concurrently by another trained laboratory scientist. The result for malaria RTD were recorded as positive or negative.
Data collection
We collected patient´s demographic information and information on associated factors such as occupation, level of education, presence of rats in the household, having a garden, and living in a flood prone area and the outcome variable using a pretested interviewer-administered structured questionnaire. The data was entered and coded in Microsoft Excel 2016. Data analysis was done using EpiInfo Software version 7.2.2
®. The main outcome variable was the presence or absence of Leptospira antibodies (either IgG or IgM or both). We coded questionnaire data and results got from the RDT as dichotomous variables for bivariate analysis. We summarized data using frequencies, proportions and median. Bivariate analysis was used to explore the association between each of the potential risk factors and the outcome variable. Level of significance was set at α0.05. Odds ratio (OR) with 95% confidence interval (CI) were used to quantify the associations.
Availability of data and materials
The data analyzed for this study is available upon proper demand from the corresponding author.
Ethical considerations
Ethical approval was obtained from the UI/UCH ethics committee (UI/EC/19/0207). Approval to work at the hospital was gotten from Oyo State Hospital Management board (OYSHMB/185 VOL IV/159). We also got verbal consent directly from participants older than and from caregivers/parents of participants below 18 years. All (malaria and leptospirosis) results obtained were documented and submitted to the physician who requested for the laboratory test. Final copy of the findings was also shared with the hospital management.
Results


The median age of the respondents was 34 years (range: 6 months to 80 years). Eighty-nine (62.2%) of the participants were females, while 91(63.6%) were unemployed. Most of the participants (62.2%) had at least a secondary education or higher, with a similar proportion (63.3%) being married Table 1.
Among the febrile patients, 60 (42%) reported having headache,38 (26.6%) had body pain, 35(24.5%) complain of stomach aches, 11(7.7%) had vomited during illness and none had jaundice
Table 2.
Of the 143 samples tested, 12 (8.4%) were positive for leptospiral IgG/ IgM. Eleven of these 12 samples were IgM positive. Eleven (7.7%) of the samples tested positive for malaria while 1 (0.7%) was positive for both Leptospira and Malaria
Table 3.
None of the factors assessed was significantly associated with having leptospirosis
Table 4.
Discussion


The results showed that the prevalence of leptospirosis among febrile patients presenting at the study center is 8.4%. This is the same as the prevalence found among febrile patients in Tanzania [17] and falls within the range of the prevalence of other studies conducted on febrile patients in other parts of Africa: Egypt (4.0-19.8%) [18, 19], Kenya (3.2-17.9%) [20], and Ghana (4.5 -7.8%) [21]. It is however much lower than the prevalence reported from the Terai region of Nepal [22]. The lower prevalence in our study could be as a result of the fact that we conducted our study between April and May when the rains were just starting as compared to the Nepal study, conducted during heavy rains and flooding [22].
Majority of the Leptospira-positive patients in this study were 25-40 years old. This is in harmony with findings from the Nepal study which also showed that persons in this age group are the active work force in the population, hence are more likely to get exposed during their various day-day jobs compared to others [
22,
23]. Sixty-six percent of those that tested positive for the disease were female. This is the same as in the Nepal study, but very different from other studies which reported higher prevalence among males [
24,
23]. This high prevalence in females may be because females are more likely to get exposed while doing household jobs like cooking and waste disposal and other household activities that may expose them to contaminated water and food material.
Also, in Ibadan, the practice of raising animals like sheep and goat is more common among women than men. The presence of Leptospira organism in farm animals in Nigeria is a well-documented fact, including in Ibadan [
1,
25]. Many studies have shown leptospirosis to be a disease of the poor and less educated in society [
22,
9]. However, this study reported a different finding in which two third of patients that tested positive for leptospirosis had a minimum of tertiary education, with an average monthly salary of approximately 28,000 naira (USD 80 at USD 1 = 350 naira), an amount above the national minimum wage of 17,000 naira (USD 49). The combination of good education and high incomes means these groups of people are more likely to have better health-seeking behaviour than the poor and less educated [
26]. This observation is, however, not statistically significant. One patient, a 3-year-old male, tested positive for both malaria and leptospirosis. That he got infected at such a young age is not abnormal because studies have reported leptospirosis in younger children in rural areas in Vietnam, where the disease is endemic [
27].
Study limitation
This is a pilot study with a relatively small sample size. We conducted the study in one facility and therefore we cannot generalize the result to the entire population of Ibadan extrapolat the results to the general population. The study was also conducted between April and May 2019 when the rainfall in Ibadan was not yet at its peak. Studies have shown that the prevalence of leptospirosis increases with increase in rainfall and subsequent flooding [
22,
28,
1,
29]. However, we have shown from this study that leptospirosis is a possible cause of acute febrile illnesses in Ibadan. Hence, there is a need for further laboratory investigations to be conducted on malaria negative samples.
Conclusion


Leptospirosis was found among febrile patients seeking medical care at the study center. We also found that the seroprevalence of leptospirosis is same as that of malaria among the sampled population. However, the frequency of leptospirosis/malaria co-infection was low. Testing for leptospirosis is therefore recommended as a differential for acute febrile illnesses.
What is known about this topic
- Leptospirosis is a highly contagious bacteria disease that can affect both humans and animals.
- The disease has a worldwide distribution but most deaths as a result of the disease occur in developing countries
- Transmission is by direct contact with infected animals or indirectly through contact with contaminated soil or water through mucous membranes or broken skin.
What this study adds
- Leptospirosis is a cause of acute fever and other malaria-like symptoms in our environment
- The prevalence of leptospirosis was shown to be the same as that of malaria among those tested
- Screening for leptospirosis can be done with RDT.
Competing interests


The authors declare no competing interest
Authors' contributions


Mathias Besong, conceived the study, Emmanuel Awosanya and Gabriel Ogundipe supervised the study, Olusoji Adeyanju handled approvals for the study, Mathias Besong and Adedayo Adigun carried out the laboratory work, Mathias Besong, Emmanuel Awosanya carried out the data interpretation, Charles Michael reviewed manuscript. Mohammed Ballogun is guarantor of the paper. All authors read and approved final version of manuscript.
Acknowledgments


I thank the staff of the laboratory unit of Adeoyo Maternity Teaching Hospital, Yemetu, Ibadan for all the hard work they put in during the study period. I am also grateful to all the participants for accepting to take part in this study. Thank you to my mentors, Dr Chukwuma and Dr Bashorun for guiding me through this process. Finally, I wish to say a big thank you to my family for their continuous support.
Tables


Table 1: Sociodemographic characteristics of febrile patients (n=143) presenting at Adeoyo Maternity Teaching Hospital, Ibadan May, 2019
Table 2: Distribution of signs/symptoms among febrile patients (n= 143) and proportion positive for Leptospira RDT (n=12), and Malaria RTD (n=11), AMTH Ibadan May, 2019
Table 3: Seroprevalence of Leptospirosis among febrile patients (n= 143) presenting at Adeoyo Maternity Teaching Hospital, Ibadan May, 2019
Table 4: Bivariate analysis of factors associated with Leptospirosis among febrile patients presenting at Adeoyo Maternity Teaching Hospital, Ibadan May, 2019 (n = 143)
References


- de Vries SG, Visser BJ, Nagel IM, Goris MGA, Hartskeerl RA, Grobusch MP. Leptospirosis in Sub-Saharan Africa: A systematic review. Int J Infect Dis. 2014;28:e47-64.https://doi.org/10.1016/j.ijid.2014.06.013 . Google Scholar
- Dobigny G, Gauthier P, Houéménou G, Choplin A, Dossou HJ, Badou S, Etougbétché J, Bourhy P, Koffi S, Durski K, Bertherat E, Picardeau M. Leptospirosis and Extensive Urbanization in West Africa: A Neglected and Underestimated Threat? Urban Sci. 2018;2:1-7.https://doi.org/10.3390/urbansci2020029 . Google Scholar
- Benacer D, Thong KL, Verasahib KB, Galloway RL, Hartskeerl RA, Lewis JW, Mohd Zain SN. Human Leptospirosis in Malaysia: Reviewing the Challenges after 8 Decades (1925-2012). Asia-Pacific Journal of Public Health. 2016 May [cited 2022 Sep 9];28(4):290–302.https://doi.org/10.1177/1010539516640350 . Google Scholar
- Borja M. Burden of Disease Study: Leptospirosis in an Urban Setting, Metro Manila. Accessed on 12 May 2012.
- Petti CA, Polage CR, Quinn TC, Ronald AR SM. Laboratory medicine in Africa: a barrier to effective health care. Clin Infect Dis. 2006;42:377-82.https://doi.org/10.1086/499363 . Google Scholar
- World Health Organization. Human leptospirosis: guidance for diagnosis, surveillance and control. Int Leptospirosis Soc Geneva. 2003. Accessed on 12 May 2012.
- Haake, DA and Levett PN. Leptospirosis in Humans. In: Leptospira and Leptospirosis. Berlin, Germany: Springer; 2015. p. 65-97. Google Scholar
- Maciel EA, de Carvalho AL, Nascimento SF, de Matos RB, Gouveia EL, Reis MG, Ko AI. Household transmission of leptospira infection in urban slum communities. PLoS Negl Trop Dis. 2008 Jan 30; 2(1):e154.https://doi.org/10.1371/journal.pntd.0000154 PubMed | Google Scholar
- Allan KJ, Biggs HM, Halliday JE, Kazwala RR, Maro VP, Cleaveland S, Crump JA. Epidemiology of Leptospirosis in Africa: A Systematic Review of a Neglected Zoonosis and a Paradigm for 'One Health' in Africa. PLoS Negl Trop Dis. 2015 Sep 14; 9(9):e000389 https://doi.org/10.1371/journal.pntd.0003899 PubMed | Google Scholar
- Agunloye CA, Alabi FO, Odemuyiwa SO OO. Leptospirosis in Nigerians: a seroepidemiological survey. Ind Vet J. 2001;78:371-5. Google Scholar
- Onyemelukwe NF. A serological survey for leptospirosis in the Enugu area of eastern Nigeria among people at occupational risk. J Trop Med Hyg. 1993; 96:301-4. Google Scholar
- CDC Division of High-Consequence Pathogens and Pathology (DHCPP). World´s Most Widespread Zoonotic Disease Poses New Risks: Focus on Leptospirosis. Medscape. 2013. Accessed Aug 2022.
- Hartskeerl RA. Current tools and emerging technologies for leptospirosis diagnosis. 2015.
- Centres for Disease Contron and Prevention (CDC). Leptospirosis Fact Sheet for Clinicians. CDC. 2018.
- Brah S, Daou M, Salissou L, Mahaman SA, Alhousseini D, B AI, Moussa S, Malam-Abdou B, Adamou H, Adehossi E. Fever of Unknown Origin in Africa: The Causes Are Often Determined! Heal Sci Dis. 2015;16:1-8. Google Scholar
- Naing, L; Winn, T; Rusli BN. Practical issues in calculating the sample size for prevalence studies. Arch Orofac Sci. 2006;1:9-14. Google Scholar
- Biggs HM, Bui DM, Galloway RL, Stoddard RA, Shadomy SV, Morrissey AB, Bartlett JA, Onyango JJ, Maro VP, Kinabo GD, Saganda W, Crump JA. Leptospirosis among hospitalized febrile patients in northern Tanzania. Am J Trop Med Hyg. 2011 Aug; 85(2):275-81. https://doi.org/10.4269/ajtmh.2011.11-0176 PubMed | Google Scholar
- Parker TM, Murray CK, Richards AL, Samir A, Ismail T, Fadeel MA, Jiang J, Wasfy MO, Pimentel G. Concurrent infections in acute febrile illness patients in Egypt. Am J Trop Med Hyg. 2007; 77:390-92. Google Scholar
- Ismail TF, Wasfy MO, Abdul-Rahman B, Murray CK, Hospenthal DR, Abdel-Fadeel M, Abdel-Maksoud M, Samir A, Hatem ME, Klena J, Pimentel G, El-Sayed N, Hajjeh R. Retrospective serosurvey of leptospirosis among patients with acute febrile illness and hepatitis in Egypt. Am J Trop Med Hyg. 2006;75:1085-89. Google Scholar
- Forrester AT, Kranendonk O, Turner LH, Wolff JW BH. Serological evidence of human leptospirosis in Kenya. East Afr Med J. 1969; 46:497-506. Google Scholar
- Janice A Tagoe, Naiki Puplampu, Shirley Cameron Odoom, Bassem Abdul-Rahman, Engy Emil Habashy, Billy Pimentel, Karl Kronmann, Kwadwo Koram, Michael Wilson, Moustapha Abdel, Edward Nyarko, Prince Agbenohevi. Serosurvey of Leptospirosis among Patients with Acute Febrile Illness in Accra. Am J Trop Med Hyg. 2010; 83(5_Suppl):306.
- Regmi L, Pandey K, Malla M, Khanal S, Pandey BD. Sero-epidemiology study of leptospirosis in febrile patients from Terai region of Nepal. BMC Infect Dis. 2017 Sep 18;17(1):628.https://doi.org/10.1186/s12879-017-2733-x PubMed | Google Scholar
- Sethi S, Sharma N, Kakkar N, Taneja J, Chatterjee SS, Banga SS, Sharma M. Increasing trends of leptospirosis in northern India: a clinico-epidemiological study. PLoS Negl Trop Dis. 2010 Jan 12; 4(1):e579.https://doi.org/10.1371/journal.pntd.0000579eCodeCamp PubMed | Google Scholar
- Panaphut T, Domrongkitchaiporn S, Thinkamrop B. Prognostic factors of death in leptospirosis: A prospective cohort study in Khon Kaen, Thailand. Int J Infect Dis. 2002; 6:52-9.https://doi.org/10.1016/S1201-9712(02)90137-2 . Google Scholar
- Jagun AT, Ajayi OL, Ilugbo MO, Olugasa BO, Kafer J SH. Isolation and prevalence of pathogenic Leptospira interrogans in slaughtered cattle in two abattoirs in southwestern Nigeria. Trib EU. 2011;1:235-7. Google Scholar
- Adam V, Aigbokhaode A. Sociodemographic factors associated with the healthcare-seeking behavior of heads of households in a rural community in Southern Nigeria. Sahel Med J. 2018; 21:31. Google Scholar
- Thai KTD, Nga TTT, Phuong HL, Giao PT, Hung LQ, Binh TQ, Van Nam N, Hartskeerl RA, de Vries PJ. Seroepidemiology and serological follow-up of anti-leptospiral IgG in children in Southern Vietnam. Acta Trop. 2008; 106:128-31.https://doi.org/10.1016/j.actatropica.2008.02.005 . Google Scholar
- Costa F, Ribeiro GS, Felzemburgh RD, Santos N, Reis RB, Santos AC, Fraga DB, Araujo WN, Santana C, Childs JE, Reis MG, Ko AI. Influence of household rat infestation on leptospira transmission in the urban slum environment. PLoS Negl Trop Dis. 2014 Dec 4;8(12):e3338.https://doi.org/10.1371/journal.pntd.0003338 PubMed | Google Scholar
- Schneider MC, Velasco-Hernandez J, Min KD, Leonel DG, Baca-Carrasco D, Gompper ME, Hartskeerl R, Munoz-Zanzi C. The Use of Chemoprophylaxis after Floods to Reduce the Occurrence and Impact of Leptospirosis Outbreaks. Int J Environ Res Public Health. 2017 Jun 3; 14(6):594. https://doi.org/10.3390/ijerph14060594 PubMed | Google Scholar