Bacteriological quality of Nono, a milk product sold at retail outlets in Federal Capital Territory, Nigeria
George Onyemauwa Uzoaga1,2,&, Chukwuma David Umeokonkwo1,3, Aishat Bukola Usman1, Grace Sabo Kia2, Emmanuel Chukwudi Okolocha2
1Nigeria Field Epidemiology and Laboratory Training Program, Asokoro, Abuja, Nigeria, 2Department of Veterinary Public Health and Preventive Medicine, Ahmadu Bello University, Zaria, Kaduna State, Nigeria, 3Department of Community Medicine, Alex Ekwueme Federal University Teaching Hospital Abakaliki, Ebonyi State, Nigeria
&Corresponding author
George Onyemauwa Uzoaga, Nigeria Field Epidemiology and Laboratory Training Program, Asokoro, Abuja, Nigeria. georgereuben2000@yahoo.com
Introduction:
Milk handling in the Nigerian informal dairy sector is commonly done without observing hygienic practices, which is a threat to food safety and public health. The indiscriminate use of antibiotics in treatment of infections in animals has led to increasing resistance of pathogenic organisms to commonly used antibiotics. We determined the bacteriological quality of nono and the antimicrobial resistance of respective bacterial isolates.
Methods:
We conducted a cross-sectional study on 300 samples of ready-to-consume locally fermented milk product (nono) sold in the Federal Capital Territory, Nigeria between June and September 2018. We used a multistage sampling technique for sample collection. Culturing on appropriate media and conventional biochemical tests were carried out for identification and isolation of bacteria of interest while further confirmatory tests were carried out using Microbact/Mocrogen® kits. Serological test was conducted using Remel RIM™ Latex to confirm presence of E. coli 0157:H7 sero-group, while Kirby-Bauer disc diffusion method was used for antimicrobial susceptibility testing.
Results:
Total Aerobic Plate Count (±SD) ranged from 5.6 ± 1.7 - 7.0 ± 0.4 log10 cfu/ml while total coliform count ranged from 5.6 ± 0.5 - 6.5 ± 0.7 log10 cfu/ml. Out of the 300 samples, 37 (12.3%) tested positive for E. coli out of which 16% were of the 0157:H7 sero-group while 21(7%) were positive for Staphylococcus aureus. E. coli isolates were found to be totally resistant to Vancomycin and Methicillin, and almost totally resistant to Ampicillin (97%) and Tetracycline (95%). Staphylococcus aureus isolates on the other hand were totally resistant to Oxacillin, Ticarcillin and Amoxycillin.
Conclusion:
Total plate counts from this study is above the maximum permissible range of 4.6 log10 cfu/ml. The bacterial isolates showed multidrug resistance varying from 3 to 8 of the antibiotics used. We recommended health education and awareness creation on hygiene practices among local milk vendors and advocated for right use of antimicrobials in animals by veterinarians.
Introduction

Raw or processed milk is a good growth medium that supports the growth of several microorganisms especially bacteria because of its high water content, nearly neutral pH, and a variety of available essential nutrients [1, 2]. As a result of the presence of these nutritional components, milk is an excellent culture medium for many microorganisms, especially bacterial pathogens [3]. These microorganisms may contaminate milk at various stages of procurement, processing and distribution. There is a constant challenge to those involved in milk production and sale to prevent or minimize the entry and subsequent growth of microorganisms in milk [4]. Bacterial contamination can generally occur from three main sources; within the udder, outside the udder and from the surface of equipment used for milk handling and storage [5]. This not only reduces the nutritional quality but also consumption of such milk threatens health of the society [4, 6].
Food borne diseases are a common and widespread global problem. Several outbreaks have been reported as a result of consuming milk that may appear normal but are in fact contaminated with large number of harmful bacteria [
7]. According to an analysis by Centers for Disease Control and Prevention (CDC), between 1993 and 2006, more than 1500 people in the United States became sick from drinking raw milk or eating cheese made from raw milk. CDC has also reported that unpasteurized milk is 150 times more likely to cause food borne illness and result in 13 times more hospitalization than illnesses involving pasteurized dairy products [
8].
E. coli is a commonly used faecal indicator organism. Its presence in food generally indicates direct or indirect faecal contamination and a general lack of cleanliness in handling and improper storage [
9].
E. coli O157:H7 infection is an emerging cause of food borne illness that causes severe bloody diarrhea, abdominal cramps and occasionally to kidney failure.
E. coli O157:H7 was first recognized as a cause of illness in 1982 during an outbreak of severe bloody diarrhea which was traced to contaminated hamburgers [
10]. Since then, most infections have come from eating undercooked beef and other foods of animal origin. An estimated 10,000 to 20,000 cases of infection occur in the United States each year [
11]. Consumers can prevent the infection by thoroughly cooking ground beef, avoiding unpasteurized milk, and washing hands carefully.
Staphyloccocus aureus is another bacterium implicated in food poisoning including milk. It multiplies in food including milk and dairy products kept at room temperature. The toxins may be present in dangerous amounts in foods that have no sign of spoilage, such as bad smell. Symptoms are nausea, vomiting retching, stomach cramping and diarrhea. In more severe cases, dehydration, headache, muscle cramping and changes in blood pressure (BP) and pulse rate may occur [
11].
With the increase of population of immuno-compromised persons in Federal Capital Territory following influx of internally displaced persons, there is increased risk of diarrhea diseases and other infections through the consumption of unregulated milk products [
12]. Bacteriological analyses are critical for the assessment of quality and safety of milk worldwide but in Nigeria and indeed most parts of Africa, there is a relative scarcity of data pertaining to the levels of pathogen in commercially available milk products and as it were to compound the situation there is non-enforcement of regulatory standards where they exist [
13]. The Standards Organization of Nigeria (SON) stipulates that Total Aerobic Plate Count for milk meant for human consumption should not exceed 50,000/ml (4.6 log
10cfu/ml) [
14] which aligns with Codex Alimentarius Standards (<5.0 log
10cfu/ml) [
15] and similar to Standard set by the Kenyan authorities (4.3 log
10cfu/ml) [
16]. A large percentage of milk is retailed directly to consumers by small-scale traders, including hawkers with virtually no quality control at all levels. In Nigeria, the Fulani and Hausa speaking tribes locally process cow milk in its raw or boiled form into products like
Nono, Kindrimo and
Manshanu [
6] and the process of preparing this milk product gives a lot of concern because of the unhygienic practices, especially the quality of water used, the processing and retailing environments. This study is therefore aimed at bacteriological analysis of locally fermented cow milk product (
Nono) sold in retail outlets in the Federal Capital Territory to determine the bacterial load, to isolate the contaminating bacteria of interest (
E. coli and
Staphylococcus aureus) and assess the antibiotic sensitivity of the bacterial isolates. The objectives of study were to determine the bacteriological quality of locally fermented cow milk product (
Nono) sold in retail outlets; to quantify the prevalence of
Escherichia coli and
Staphylococcus aureus from the samples; to determine what proportion of isolated
Escherichia coli that is of the 0157:H7 sero-group and to determine antibiotic sensitivity of the bacterial isolates to commonly used antibiotics.
Methods


Study setting, design and sample size
The Federal Capital Territory is made of six area councils - Abaji, Bwari, Kwali, Kuje Gwagwalada and Abuja Metropolitan Area Council (AMAC). Each of the area councils has an administrative headquarters and is governed by elected officials. The area houses the countries administrative capital with almost all the headquarters of important government Ministries, Departments and Agencies located with the area. It is largely a cosmopolitan area with all tribes and people represented. It still has a sizeable proportion of the locals on the outskirts of the territory.
The area is a rapidly developing with a lot of construction works ongoing all-round the year. Around these construction sites and in many other areas in the sites there are small retail shops set up to meet the needs of the workers, and other people that have visited for different purpose.
We conducted a cross sectional study among selected vendors of locally fermented cow milk in the Federal Capital Territory, Abuja, Nigeria. Using Lemeshow & Lwanga formula for cross sectional studies [
17], a sample size of 300 was obtained after adjusting for 10% non-response rate.
Sampling Technique
We used multi-stage sampling technique to select samples for the study. In the first stage, two districts were selected by balloting from each Area Council from the list of the districts in each district. In stage two, five catchment areas were selected from each district by balloting giving a total of 60 catchment areas. In stage three, in each catchment area, five
nono samples were collected from the vendors as they arrived the retail outlets giving a total 300 samples.
Specimen Collection Methods and Analysis
A total of 300 raw milk samples (about 25ml each) were collected at selling points into sterile corked plastic tubes and labeled appropriately with date and time of collection recorded and an identification number assigned to each sample. Samples were placed in separate sterile plastic bags to prevent spilling and cross contamination and stored in a cooler with frozen ice packs before being transported in an unbroken cold chain (4±2°C) to the Bacterial Zoonoses Laboratory of the Department of Veterinary Public Health and Preventive Medicine, Ahmadu Bello University Zaria, for Laboratory analysis.
Laboratory Analysis
Total aerobic plate count and total coliform count were carried out using nutrient agar and MacConkey agar (Oxoid) respectively while EMB and MSA were used for isolation of
E. coli and
Staphylococcus aureus respectively after culturing at 37 °C for 18 to 24 hours. Media used for biochemical analysis included: Triple sugar iron (TSI) agar, Sulphur indole motility (SIM) medium, urea agar base +40% urea solution, Simmons citrate agar and Methyl red-Voges Proskauer medium.
The bacteria were identified by colonial characteristics, Gram´s staining differential and selective media, Sugars and conventional biochemical tests [
18]. Further confirmation was carried out with Microbact and Microgen Kits for
E. coli and
Staphylococcus aureus isolates respectively. The Microbact (for
E. coli isolates) and Microgen tests (for
Staphylococcus aureus isolates) were carried out according to the Manufacturer's instructions (Oxoid Ltd). Serological test was conducted using Remel RIM
™ E. coli O157:H7 Latex Test to identify the percentage of the
E. coli isolates that are of the serogroup O157:H7. Test includes 3 latex reagents coated with a different antibody: one against
E. coli serotype O157, another against
E. coli serotype H7, and the third with normal rabbit globulin, to serve as the control. Agglutination confirms presence of
E. coli O157:H7. No agglutination indicates the test isolate is not
E. coli O157:H7 while Control Latex reagent identifies non-specific agglutination.
Determination of Antimicrobial Susceptibility Pattern
Antibiotic sensitivity test was carried out using the Kirby-Bauer disc diffusion method (Bauer et al.,) [
19]. The isolates were sub-cultured on selective media. The overnight culture broths were adjusted to match 0.5 Mc Farland turbidity standard. Then the prepared Mueller Hinton agar were swabbed and allowed to stand for 10 minutes. Antibiotics were applied on the swabbed Mueller Hinton agar plates using antibiotic dispenser machine, then incubated at 37°c for 24 hours.
The zones of inhibition were measured in mm using a transparent ruler. Results were interpreted using Interpretive Standards of the Clinical and Laboratory Standards Institute (CLSI), 2008 Edition [
20] and also to classify the various antimicrobials tested as resistant (R), intermediate (I) or sensitive (S) based on diameter of zones of inhibition measured to the nearest millimeter. Multiple antibiotics resistance was defined as resistance to three or more classes of antimicrobial agents.
Ethical Clearance
Ethical clearance was obtained from the Ethics Committee, Federal Capital Territory Health and Human Services, Abuja with approval number: FHREC/2017/01/53/16-06-17
Data Analysis
Data were analyzed using Excel and SPSS Software packages and results presented in frequencies, proportions and charts.
Results


The mean log Total Aerobic Plate Count (TAPC), (±SD) ranged from 5.6 ±1.7 log10 cfu/ml (AMAC) to 7.0 ± 0.36 log10 cfu/ml (Kuje). The mean log Total Coliform Count (TCC) ranged from 5.6 ± 0.5 to 6.5 ± 0.7 log10 cfu/ml Table 1. The overall prevalence of pathogenic E.coli was 12.3% and 7% for Staphylococcus aureus. The prevalence of isolated E. coli (3.67%) was higher in the Gwagwalada area councils than other area councils and least in Abaji area council (1%). The prevalence of S. aureus was highest in Kwali area council (2.3%) and least in Kuje area council (1%). All the samples contained microbial counts (Total aerobic plate counts and Total Coliform counts) above the maximum permissible of 4.6 log10 cfu/ml.
Of the 300 samples evaluated, (after plate culturing/incubation at 37oc for 24 hrs), 104 (34.7%) were positive for
E. coli (Green metallic sheen on EMB Agar) while 83 (27.7%) samples showed the characteristic golden yellow growth of
Staphylococcus colonies on MSA medium
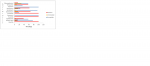
Figure 1: Antimicrobial susceptibility pattern of E. coli isolates from Nono - fermented milk product sold in FCT, Abuja.
Isolation and Characterization of E. coli and Staphylococcus aureus
At first culturing on appropriate media, 104 and 83 samples were positive for
E. coli and
S. aureus respectively. When subjected to Conventional Biochemical and Sugars tests, 54 isolates were confirmed to show typical
E. coli reactions while only 25 tested positive for
Staphylococcus aureus. (These were taken as presumptuous isolates). Further confirmatory tests with Microbact
™/Microgen
™ Systems for
E. coli and
S. aureus respectively, yielded 37 (12.3%) positives for
E. coli and 21 (7%) for
Staphylococcus aureus Table 2.
Isolation of E. coli 0157:H7
Result of serological test (Remel RIM
™ E. coli O157:H7 Latex) showed that 16 (43%) of the 37
E. coli belonged to the 0157:H7 sero-group by testing positive to the latex agglutination test. This constitutes 5.3% of the total survey sample (contaminated by
E.coli 0157:H7).
Result of Antimicrobial sensitivity tests
Among the 13 antibiotics used for antibiotic susceptibility test, the
E. coli isolates were found to be totally resistant to Vancomycin (100), Cefixine (100%), Methicillin (100%), Ticarcillin (100%) and almost totally resistant to Ampicillin (97%) and Tetracycline (95%).
Staphylococcus aureus isolates on the other hand were totally resistant to OxaciFllin (100%), Ticarcillin (100%) and Amoxycillin (100%). All (100%) of the isolates - both
E. coli and
Staphylococcus aureus showed multi-drug resistance varying from 3 to 8 of the antibiotics
Figure 2,
Figure 3.
In terms of susceptibility, the
E. coli isolates were totally susceptible to Azithromycin (100%), and Nalidixic acid (100%) but not totally susceptible to Imipenem (91.8%) while
Staphylococcus aureus isolates were also 100% susceptible to Kanamycin, Gentamycin and Imipenem respectively.
Discussion


Our study showed that the bacteriological level of locally fermented milk in the Federal Capital Territory, Nigeria is above the standards set by Codex Alimentarius on fermented milk products [15] as well as the maximum permissible bacterial count recommended by the Standards Organization of Nigeria (SON)[14] and also above the European Commission Standard [21]. Greater levels of bacteria and coliform counts such as those reported here have been reported in different places in Tanzania [22] and in other African countries [23]. The short-term solution for consumers at this moment is continuation of boiling milk before consumption and avoidance of taking raw milk traditionally processed products such as fermented milk, ghee, and local yoghurt. Concerns of consumers being exposed to different forms of milk hazards and associated risks have been reported regularly in the last two decades [24]. Evidences gathered in this study indicate that contamination and nutritional risks through consumption of unpasteurized milk remains a reality in our environment - Abuja, and Nigeria in general especially pertaining to the isolation of E.coli 0157:H7 which causes bloody diarrhea and other complications such as hemolytic uremic syndrome (HUS) leading to acute renal failure. Staphylococcus aureus on the other hand produces enterotoxins and has many ill effects on humans including, for example, infertility in males and females [25] and has been associated with mastitis in animals [26]. E.coli species reported in high numbers in many countries represent a threat to food safety and are mainly contributed by fecal contamination from ruminants [27]. Although E. coli is frequently occurring organisms in milk and its products, the incidence of the pathogenic species of E. coli itself in milk and milk products as a possible cause of food borne disease is becoming a matter of public health concern.
The high levels of the bacterial load in the milk as seen from this study may be attributed to unhygienic practices during milking of cows and in the process of preparation up to the point of sale of the milk product by the vendors. This finding of high possibility of bacterial contamination is in agreement with Eka and Ohaba [
28], who observed that the vendors who produce and market this locally fermented milk (nono) do not observe any standard hygienic procedures in the preparation of this product and that the failure of most of these handlers to pasteurize their milk also results in high TAC as pasteurization reduces the microbial load of food to safer levels.
Aerobic count which indicates microbial quality of food products is useful as an indicator for the food quality and shelf life (Pianetti
et al., 2008). The overall TAC level observed in this study was lower than the mean log bacteria counts by Makut
et al., in Keffi, Nasarawa State, Nigeria [
6]. Lawan
et al. (2012) reported slightly lower counts in raw milk from two dairy farms in Zaria, Nigeria as 5.70 and 6.04. Ogbonna
et al [
29] reported an overall mean of 5.34 from raw milk in Maiduguri while Laba and Udosek [
30] reported a range of 6.04 - 6.41 for raw milk in Ilorin, Nigeria. Tamba
et al., (2016) [
31] reported a higher mean value of 7.22 in fermented milk from Zaria, Kaduna State, Nigeria. Okonkwo (2011) [
29] reported a range of log 0 to 8.9 TAC for fermented milk from different markets in Maiduguri, Nigeria. This finding partly disagrees with Egwaikhide
et al. (2014) [
32] who reported mean TAC as low as 3.48 to TNTC for fermented milk (nono) consumed within Kaduna, Nigeria.
The results of the serological test conducted using Remel RIM
™ Latex Test which showed that 5.3% of the samples were contaminated by
E. coli 0157:H7 is similar to the work of Reuben
et al; (2013) [
33] who reported an
E. coli 0157:H7 prevalence of 4.5% in
nono milk in Nasarawa State, Nigeria.
Milk contamination to unacceptable levels in other studies and in this present study can be attributed to the fact that poor hygienic practices and poor management systems are common among the milkers as well as the retailers in virtually all parts of the country. Poor hygiene practices by milk handlers leads to the introduction of pathogenic microorganisms into the milk [
28].
On the problem of drug resistance, both
E. coli and
Staphylococcus aureus isolates showed multi drug resistance to commonly used antibiotics. This result is in agreement with the findings by other researchers, who reported multidrug resistance among
E. coli isolates. Worthy of mention is the development of resistance of
E. coli to Imipenem to which it is known to be susceptible. This result can be attributed to the indiscriminate administration of antibiotics especially of sub therapeutic doses which renders the organisms´ drug-tolerant while drug residues in milk also makes consumers drugs tolerant especially when the withdrawal time before slaughter is not observed in food animals. Evidence has been found which indicates that resistant strains of pathogens can be transmitted to humans through food [
34]. Results from this study indicate that Azithromycin), Nalidixic Acid and Imipenem, are the drugs of choice for
E. coli, since none of the isolates was resistant to them. Cephtazidine was also effective (70%) against
E. coli pathogens which is slightly lower than (80%) recorded in a study by Reuben
et al, (2013)[
35].
Staphylococcus aureus isolates were totally resistant to Amoxycillin, Oxacillin and Ticacillin while on the other hand they were susceptible to Kanamycin (100%), Gentamycin (100%), Imipenem (100%), Nalidixic Acid (90%) and Chloramphenicol (86%) respectively. Thus, veterinarians and clinicians alike should prioritize the use of these effective drugs in treating Staphylococcus infections in animals and humans as the case may be while avoiding those to which the organism has developed resistance.
The public health significance of these findings is that antimicrobial resistant
E. coli (especially O157:H7) and
S. aureus from 'nono' (or dairy animals) may colonize the human population via the food chain, contact through occupational exposure, or waste run off from 'nono' production facilities to the neighborhood. Indiscriminate use of antimicrobials among livestock producers and marketers in Nigeria could also be responsible for the resistance pattern obtained in this study.
This is in agreement with the finding of Olatoye (2010) [
36], which recorded a high level of tetracycline resistance of 91.4% among isolates of E. coli O157:H7. In Malaysia, Al Haj (2007)[
37] also observed high resistance to tetracycline (81.4 %) while Shitandi (2001) also obtained high resistance to penicillin (72%) and to Tetracycline (57.9%) [
35].
The high level of resistance of tetracycline obtained in this study may be as a result of it being the most commonly available antibiotic used as growth promoter and routine chemoprophylaxis among livestock in Nigeria. This is worrisome considering that tetracycline is a first line drug in Nigeria, and as in most developing countries, people with gastrointestinal infections readily purchase it across the counter for self-medication or treatment of animals. In addition, the high prevalence of resistance of
E. coli isolates to Vancomycin, Ampicillin, Methicillin, Tcacillin and Cefixime is of importance from the view point of medical and veterinary practice in Nigeria. This could be a reflection of use and misuse of these antibiotics in the society.
Conclusion


This study has shown the overall bacterial load of fermented cow milk sold in the FCT, Abuja are of poor quality and are contaminated by pathogenic E. coli and Staphylococcus aureus bacteria. This may be attributed to lack of effective sanitary precautions and less careful handling procedures during milking process and nono production. The use of traditional milking methods also exposes milk to pathogenic bacteria found in cow udders and probably on the hands of the milkers who may have come in contact with faeces of the cows or that of their young children. The unhygienic environmental conditions where nono is marketed may have also contributed to its contamination.
The development of antimicrobial resistance by the bacteria pose a major challenge in both human and animal medicine because these drugs are commonly used in the treatment of human patients and in veterinary practice and antibiotic resistance can be developed through consumption of unpasteurized milk contaminated by these bacteria. This finding is not surprising as population have easy access to various antibiotics at any drug store without prescription from a medical or veterinary practitioner.
The detection of
E. coli and
Staphylococcus aureus in locally-fermented milk ('nono') sold in the Federal Capital Territory, Nigeria suggests that 'nono' consumption has potential health risks to consumers in the Territory. The data suggest that the indiscriminate use of these antibiotics whether therapeutically in human and veterinary medicine or as prophylaxis in the animal production, is a key driving force in the selection of antimicrobial resistance by these pathogens. It is therefore recommended that: Public Health Department of the MFCT and the Area Councils should educate all vendors of locally processed milk products within the FCT on the necessity of hygiene in the fermentation, proper handling, transportation, and adequate covering of their products to minimize contamination by ubiquitous microorganisms. Area Council Health Authorities should set up systems to periodically collect samples of this product (nono) for laboratory testing to ensure that the bacterial load is within the acceptable limits for human consumption and regulations pertaining to prohibition of purchase of antibiotics used by both humans and animals, over the counter without doctor's prescription should be enforced in the country, Nigeria.
What is known about this topic
- 'Nono' is a locally fermented milk produced by the Fulani speaking tribe of Nigeria. The process of preparing this milk product gives a lot of concern because of the unhygienic practices, the poor water quality as well as the poor processing environment.
- Various bacteria of public health concern have been reported in milk and dairy products worldwide such as Salmonella spp., Listeria monocytogenes, Campylobacter jejuni, pathogenic strains of Escherichia coli and enterotoxigenic strains of Staphylococcus aureus but in Nigeria and indeed most parts of Africa, there is hardly any checks conducted to ascertain the bacteriological quality of vended food milk products and there is relative scarcity of data pertaining to the levels of pathogen in commercially available milk products and there is non enforcement of regulatory standards.
What this study adds
- The bacteriologic quality of a milk product (Nono) sold at retail outlets in the Federal Capital Territory of Nigeria was determined and found to be below the minimum standard recommended by the Standards Organization of Nigeria and 5.3% of the tested samples were contaminated by E. coli 0157:H7 similar to the work of Reuben et al carried out in Nasarawa State, Nigeria.
- The antimicrobial resistance by the isolated bacteria in this study to commonly used antibiotics in human patients and veterinary practice pose a major challenge. Thus veterinarians and clinicians alike should prioritize the use of these effective drugs in treating Staphylococcus and E. coli infections in animals and humans as the case may be while avoiding those to which the organism has developed resistance.
Conflicts of interest


The authors declare no competing interests.
Funding
This study was supported financially by the United States Centers for Disease Control and Prevention through African Field Epidemiology Network to the Nigeria Field Epidemiology and Laboratory Training Programme. Its contents are solely the responsibility of the authors and do not necessarily represent the official views of the United States Centers for Disease Control and Prevention or the Department of Health and Human Services.
Authors Contribution


GOU conceptualized the work, designed the study, acquired data, conducted the laboratory investigation, drafted the initial manuscript. GSK and ECO supervised the laboratory investigation, contributed to the design of the work
CDU, ABU, GSK and ECO made substantially reviewed the manuscript for intellectual content,
All the authors reviewed, read and approved the final draft.
Tables and figures


Table 1: Mean Value of Bacterial Load of fermented milk samples
Table 2: Occurrence of E. coli and Staphylococcus aureus in locally-fermented milk (Nono) in the Federal Capital Territory, Abuja
Figure 1: Antimicrobial susceptibility pattern of E. coli isolates from Nono - fermented milk product sold in FCT, Abuja
Figure 2: Antimicrobial susceptibility pattern of Staphylococcus aureus isolates from Nono - fermented milk product sold in FCT, Abuja
Figure 3: Bacterial load of 300 fermented milk samples taken from retail outlets in the Federal Capital Territory, Abuja
References


- Reta MA, Addis AH. Microbiological Quality Assessment of Raw and Pasteurized Milk. International Journal of Food Science and Microbiology. 2015; 2(6): 87-9. Google Scholar
- Abate M, Wolde T, Niguse A. Bacteriological Quality and Safety of Raw Cow´s Milk in and around Jigjiga City of Somali Region, Eastern Ethiopia. International Journal of Research Studies in Biosciences (IJRSB). 2015; 3(5):48-55. Google Scholar
- Saeed A, El Zubeir IE, El Owni O. Antimicrobial Resistance of Bacteria Associated with Raw Milk Contaminated by Chemical Preservatives. World Journal of Dairy & Food Sciences. 2009; 4(1): 65-69. Google Scholar
- Oladipo IC, Tona GO, Akinlabi EE, Bosede OE. Bacteriological quality of raw cow´s milk from different dairy farms in Ogbomoso , Nigeria. Int. J. Adv. Res. Biol. Sci. 2016; 3(8):1-6. Google Scholar
- Belbachir C, Khamri M, Saalaoui E. Microbiological quality of the raw cow milk at three rural communes of the eastern region of Morocco. International Food Research Journal. 2015; 22(4):1675-1680. Google Scholar
- Makut MD, Nyam MA, Amapu TY, Ahmed, AM. Antibiogram of bacteria isolated from locally processed cow milk products sold in Keffi metropolis, Nasarawa state, Nigeria. Journal of Biology, Agriculture and Healthcare. 2014; 4(4):19-25. Google Scholar
- Varga, L. Microbiological quality of commercial dairy products: A Research Report. Formatex Microbiology Series. 2007. Google Scholar
- Centres for Disease Control and Prevention. Estimates of Foodborne Illness in the United States. CDC. 2009.
- Microbiological Guidelines for Food: For ready-to-eat food in general and specific food items. Centre for Food Safety. 2014. Accessed February 2020.
- CDC. Preventing Foodborne Illness: Escherichia coli O157:H7. CDC. 1993. Accessed April 2019.
- USA Food and Drug Administration. What You Need to Know about Foodborne Illnesses. 20 Accessed March 2018.
- Microbiology. Accessed March 2018.
- NAFDAC. NAFDAC Mandate. NAFDAC. Accessed March 2019.
- Standards Orgaanisation of Nigeria. Nigerian Industrial Standard, NIS, Standard For Liquid Milk Part 1. 1996; pg 1-7.
- FAO/WHO. Codex Standard for Fermented Milks - CODEX STAN 243-2003. Codex Alimentarius. 2011; pg 56-58.
- Technical committee. Raw whole goat milk – Specification. Kenya Bureau of Standards. 2016.
- Pourhoseingholi MA, Vahedi M, Rahimzadeh M. Sample size calculation in medical studies. Gastroenterol Hepatol Bed Bench. 2013; 6(1):14-17. PubMed | Google Scholar
- Barrow G I, Feltham RKA. Cowan and Steel´s manual for the identification of medical bacteria (3rd edition). 1993. United Kingdom. Cambridge University Press. Google Scholar
- Bauer AW, Kirby WM, Sherris JC, Turck M. Antibiotic susceptibility testing by a standardized single disk method. Am J Clin Pathol. 1966; 45:149-158.https://doi.org/10.1093/ajcp/45.4_ts.493. Google Scholar
- Wikler MA, et al. Performance Standards for Antimicrobial Susceptibility Testing, Eighteenth Informational Supplement. Clin Lab Stand Inst. 2008; 28(M100-S): 1-18. Google Scholar
- European Parliament and the council of the European Union. Laying down specific hygiene rules for on the hygiene of foodstuffs. Official Journal of the European Union. 2004;1:69-74.
- Joseph, E. Assessment of microbiological hazards along the milk value chain in Kilosa and Mvomero districts, Tanzania. Sokoine University of Agriculture Morogoro, Tanzania. 2015. Google Scholar
- Jabbar MA, Grace D, et al. Regulations for safety of animal source foods in selected Sub-Saharan African countries: Current status and their implications. Prep Safe Food, Fair Food Proj Int Livest Res Institute, Nairobi, Kenya. 2012.http://dx.doi.org/10.22004/ag.econ.181867 Google Scholar
- Dhanashekar R, Akkinepalli S, Nellutla A. Milk-borne infections. An analysis of their potential effect on the milk industry. Germs. 2012 Sep; 2(3):101-109.https://doi.org/10.11599/germs.2012.1020 PubMed | Google Scholar
- Ugwa EA, Ashimi A, Abubakar M, Obadire S. Poor semen parameters among infertile couples presenting at a gynaecological clinic of Federal Medical Centre Birnin Kudu North-west Nigeria. Niger Med J. 2015 Jul-Aug; 56(4): 283-286.https://doi.org/10.4103/0300-1652.169746. PubMed | Google Scholar
- Anueyiagu KN, Isiyaku AW. Isolation, identification of Staphylococcus aureus from bovine milk and its antibiotics susceptibility. International Jornal of Livestock Production 2015 June; 6: 74-77.https://doi.org/10.5897/IJLP2015.0248.
- Escherichia coli (E. coli) as Indicator of Fecal Contamination. Bacteria Facts. Accessed May 2018.
- Uzeh RE, Ohenhen RE, Rojugbokan AK. Microbiological and Nutritional Qualities of Dairy Products- Nono and Wara. Nature and Science. 2006 Jan; 4(3). Google Scholar
- Innocent Okonkwo O. Microbiological Analyses and Safety Evaluation of Nono: A Fermented Milk Product Consumed in Most Parts of Northern Nigeria. International Journal of Diary Science. 2011; 6:181-189.https://doi.org/10.3923/ijds.2011.181.189. Google Scholar
- Laba SA, Udonsek CE. Bacteriological Quality and Safety Evaluation of Raw Cow Milk in Ilorin, North Central Nigeria. Nature & Science. 2013 Oct; 11(10):73.
- Wanjala WN, Nduko JM, Mwende MC. Coliforms Contamination and Hygienic Status of Milk Chain in Emerging Economies. Journal of Food Quality and Hazards Control. 2018; 5(1): 3-10. https://doi.org/10.29252/jfqhc.5.1.3. Google Scholar
- Afolabi LO. Assessment of the Microbiological Qualities of Locally and Industrially Produced Yoghurt in Lagos. Dutse Journal of Pure and Applied Sciences (DUJOPAS). 2017 June; 3(1). Google Scholar
- Reuben, CR, Okolocha EC, Bello M, Tanimu H. Occurrence and Antibiogram of Escherichia Coli O157:H7 in Locally Fermented Milk (Nono) Sold Under Market Conditions in Nasarawa State, Nigeria. International Journal of Science and Research (IJSR). 2013 Feb; 2(2). Google Scholar
- Shitandi A, Sternesjö Å. Detection of Antimicrobial Drug Residues In Kenyan Milk. Journal of Food Safety. 2001 Dec; 21(4):205-214.https://doi.org/10.1111/j.1745-4565.2001.tb00319.x. Google Scholar
- Reuben RC, Owuna G. Antimicrobial Resistance Patterns Of Escherichia Coli O157:H7 From Nigerian Fermented Milk Samples In Nasarawa State, Nigeria. International Journal of Pharmaceutical Science Invention. 2013 Mar; 2(3):38-44. Google Scholar
- Olufemi I, Olatoye IU. The incidence and antibiotics susceptibility of Escherichia coli O157: H7 from beef in Ibadan Municipal , Nigeria. African Journal of Biotechnology. 2010 Feb; 9(8):1196-1199.https://doi.org/10.5897/AJB09.1447. Google Scholar
- Alhaj N, Mariana NS, Raha AR, Ishak Z. Prevalence of Antibiotic Resistance among Escherichia coli from Different Sources in Malaysia. International Journal of Poultry Science. 2007; 6(4):293-297. https://doi.org/10.3923/ijps.2007.293.297. Google Scholar