Trends and spatial distribution of Human African Trypanosomiasis (HAT), Uganda, 2005 – 2015: A descriptive analysis of surveillance data
Susan Kizito1,2,&, Charles W. Wamboga2, Edridah Muheki Tukahebwa2, Abbass Kakembo2, Lilian Bulage1,3, Alex Riolexus Ario1
1Uganda Public Health Fellowship Program, Kampala, Uganda, 2Neglected Tropical Diseases Program- Ministry of Health, Kampala, Uganda, 3African Field Epidemiology Network, Kampala, Uganda
&Corresponding author
Kizito Susan, Uganda Public Health Fellowship Program, Kampala Uganda
Email: kizitosusan@musph.ac.ug
Background:
Human African Trypanosomiasis (HAT), commonly referred to as sleeping sickness, is a neglected tropical disease endemic in Sub-Saharan Africa. It is caused by Trypanosoma brucei (T.b.) sub-species: rhodesiense (Rhodesian HAT) and gambiense (Gambian HAT) parasites. The World Health Organization (WHO) targeted HAT for elimination as a public health problem by 2020 (annual incidence ≤1/10,000, and early diagnosis for all cases). We evaluated progress towards HAT elimination in Uganda and described its epidemiological characteristics.
Methods:
We analysed data reported from sleeping sickness treatment centres (SSTCs) to the Uganda National Sleeping Sickness Control Program during 2005–2015. A HAT case was defined as identification of T.b. rhodesiense or T.b. gambiense parasites in the blood (early-stage) or cerebrospinal fluid (late-stage) of patients at a SSTC. We analysed the cases’ distribution by person, time, and place. We used projected population data from 2002 and 2014 censuses, and linear regression to evaluate temporal trends.
Results:
During 2005–2015, SSTCs reported 2,032 HAT cases, (55% Rhodesian and 45% Gambian). The number of districts reporting AT reduced from 18 in 2005 to three districts in 2015. The average annual incidence rate (/10,000) during 2005–2015 was similar between males (Gambian=0.1, Rhodesian=0.76) and females (Gambian=0.09, Rhodesian=0.74). Gambian AT incidence declined annually by 24% (IRR=0.76; CI: 0.74-0.78) while Rhodesian AT declined annually by 13% (IRR=0.87; CI: 0.85-0.89). However, for both diseases more than 50% of cases were diagnosed in the late stage.
Conclusion:
Uganda has achieved the WHO 2020 elimination target for AT incidence. However, many cases are still diagnosed late. We recommend sustaining efforts to achieve eradication, emphasising early case detection and diagnosis and follow up of all AT cases to ensure good treatment success.
Introduction

Human African Trypanosomiasis (HAT), commonly referred to as sleeping sickness, is one of the epidemic-prone neglected tropical diseases (NTD) in Uganda [1,2]. It is caused by the protozoan parasite Trypanosoma brucei (T.b.) sub-species: T.b. rhodesiense (rhodesiense HAT) and T.b.gambiense (gambiense HAT). It is transmitted by an infective bite of tsetse flies of the genus Glossina [3], and mainly affects the rural poor. Gambiense HAT, which is primarily transmitted person-to-person, has an incubation period ranging from months to years, while rhodesiense HAT, a zoonotic disease with cattle as the main reservoir, has an incubation period of one to three weeks [4]. Both gambiense and rhodesiense HAT initially present with similar non-specific symptoms such as skin chancre, intermittent fever, headaches, enlarged lymph glands, extreme fatigue, muscle and joint pain in the early stages [4,5].
Diagnosis and treatment of the disease is complex and requires a committed and skilled workforce [
1,
4]. A definitive diagnosis is made after confirming presence of trypanosomes in either skin chancre, blood, or lymphatic gland fluid, which is referred to as early-stage diagnosis, or the presence of trypanosomes in cerebral-spinal fluid (CSF), referred to as late-stage diagnosis [
1,
4,
6,
7]. There are two laboratory methods for diagnosis of HAT depending on the sub-type. Microscopy is used for rhodesiense HAT, while the card agglutination test for trypanosomiasis (CATT) is used for gambiense HAT. However, diagnosis could be delayed or even complicated by the possible overlap of parasites [
8]. Late-stage disease is characterised by the presence of central nervous system symptoms and signs such as insomnia, progressive mental confusion, slurred speech, behavioral changes, and difficulty walking.
If untreated, HAT is fatal [
7]. Treatment depends on the clinical stage of the disease [
1]. The drugs used in the early stage of disease are safer and easier to administer, and prognosis is better. Treatment success depends on the ability of the drugs to quickly cross the blood-brain-barrier [
7]. Late stage treatment for both HAT sub-types uses melasoprol, but among gambiense HAT cases, treatment failure with melasoprol has been observed to increase [
8]. The number of treatment failures and poor outcomes among gambiense HAT cases may be increasing due to treatment associated with late stage of HAT disease. In addition, poor treatment outcomes among HAT cases could be due to an overlap during diagnosis leading to admission of inappropriate treatment of HAT patients [
8].
The World Health Organization (WHO) provides free HAT drugs to endemic countries such as Uganda [
1], and has set a 2020 target of eliminating HAT ‘as a public health problem’, defined as ≤1 HAT case/10,000 population, and early diagnosis of all HAT cases [
4,
9].
Uganda is the only African country with both
rhodesiense and
gambiense HAT, which exist in 2 geographically distinct foci: the South Eastern (SE) focus, which primarily has
rhodesiense HAT, and the North West (NW) focus, which primarily has gambiense HAT [
4,
10]. Since 2000, Uganda’s HAT control program has relied primarily on active and passive surveillance for disease, and vector control [
11,
12]. Prior to 2005, HAT incidence in Uganda was not clearly documented because
gambiense HAT surveillance had not been initiated although
rhodesiense HAT surveillance had been initiated in 2000. However, it had been observed that the land area covered by disease and the population at risk of disease were historically increasing [
8]. In addition, there was an outbreak of
T.b. rhodesiense HAT in eastern Uganda between 2001 and 2005 [
13].
Although there has been a decline in HAT incidence during the last five years in the African region, including in Uganda, the influx of refugees from both the neighboring Democratic Republic of Congo (DRC) and Southern Sudan, occurring since the year 2000, increases Uganda’s risk of new HAT infections [
11,
12,
13]. Infected refugees may act as reservoirs for the vectors, challenging national efforts to achieve elimination targets [
16].
As an additional challenge to elimination targets, during 2005-2015, African countries only infrequently reported on diagnostic stage (early or late) and treatment outcomes of HAT patients. Generally in the African region, national reports of HAT cases greatly underestimate actual HAT cases, and about 30% of the HAT cases in Africa die undiagnosed [
17]. To inform current progress towards disease elimination, we described the epidemiological trends of HAT incidence, disease stage at diagnosis, and treatment outcomes of HAT patients reported in the Uganda National Sleeping Sickness Control Program (NSSCP) database from 2005 to 2015.
Methods


Study setting: We conducted the study in Uganda, a country with a population of 37 milion people primarily living in rural settings [18]. Uganda is neigbored by two highly HAT-endemic countries, including the DRC to the west, and South Sudan to the north, and currently receives a substantial number of refugees from South Sudan and DRC.
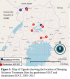
Study design: We conducted a retrospective cross-sectional study reviewing routinely-collected data reported through the NSSCP database at the Vector Control Division of Ministry of Health (VCD-MOH) from 2005-2015. The NSSCP database is fed by the 22 designated sleeping sickness treatment centers (SSTC) in Uganda, reporting patients from 20 districts in 2005 and 22 districts in 2015. There are 22 authenticated SSTC located in the endemic districts of northern and eastern Uganda. Seventeen of the SSTC reported
rhodesiense HAT cases and five reported
gambiense HAT cases in Uganda during this time period (
Figure 1).
Human African Trypanosomiasis surveillance system in Uganda: The surveillance system for HAT primarily relies on identification and treatment of HAT cases (
Figure 2). The surveillance system is based within districts with disease foci. Surveillance for HAT is done either through passive screening or active screening. Passive screening involves the patient seeking health care in the health facility while active screening involves an initiative of healthcare workers to conduct mass community screenings among populations at risk [
5]. Neither patients nor healthworkers commonly think of HAT infection, and many patients are mis-diagnosed, resulting in underreporting [
17]. Initially, active screening uses rural-friendly rapid diagnostic tests, and persons who test positive are subjected to microscopic confirmation of trypanosomes in body fluids. The rapid diagnostic test requires only a small quantity of blood and can easily be performed at the community level, producing results within 15 minutes [
5].
Demographic and clinical details of confirmed HAT patients are captured in health facility registers upon reception. Because of the nonspecific nature of HAT symptoms in the early stage, clinicians need to have a high index of suspicion to diagnose the infection. When attending to a patient with suspected HAT, the clinician will fill a laboratory request for HAT diagnosis. A tissue sample from either a chancre, blood or cerebral spinal fluid is taken for laboratory screening. Laboratory-confirmed HAT patients are recorded daily at each of the 22 SSTCs.
We defined a confirmed HAT case as a laboratory-confirmed
Trypanosoma brucei rhodesiense or
Trypanosoma brucei gambiense parasites in either a chancre, blood, or CSF of a patient being treated at one of the 22 authenticated SSTC in Uganda during January 2005 to December 2015. An electronic copy of the patient data is routinely created from the registers using Microsoft Excel and saved on the treatment facility computer. This dataset is then reported monthly to the NSSCP database by the health facility HAT focal person and archived (
Figure 2).
Data abstraction: We abstracted data reported on both
gambiense and
rhodesiense HAT cases for the period (2005-2015), SSTC, and district from the NSSCP database at the VCD-MoH offices. Even though Rhodesian HAT data started in 2000 and Gambiense HAT data started in 2005, only data on both HAT sub-types in the period 2005-2015 were extracted.
Study variables: For each of the confirmed HAT cases, we abstracted data variables of age, sex, village, parish, sub-county, district, treatment centre, diagnostic stage, date of admission, treatment and outcome. We also extracted population data from the Uganda National Census 2002 and 2014 [
19,
20] to calculate incidence.
Data management and analysis: We merged and cleaned data for each HAT sub-type using Microsoft Excel 2010. We conducted statistical analyses using Epi Info version 7.2. We conducted descriptive analysis of the HAT cases data by time, place, and person. We conducted univariate analysis to obtain frequencies, proportions of age, sex, treatment outcomes for each disease sub-type. During analysis, we categorised the age by three standardised age-groups (<5, 5-14, and ≥15 years old) and calculated disease incidence by age-group and per district using projected population data from the 2002 and 2014 census [
19,
20]. We calculated disease incidence with the number of cases as a numerator, and the population at risk per affected district as the denominator. We used Quantum Geographic Information Sysem (QGIS) version 2.8.2 to generate maps. Annual populations were estimated using a growth rate of 3.03%. The incidence (per 10,000 population) was calculated using the number of cases as the numerator and the total distict population as the denominator. Geographic differences in incidence by sub-type across districts were illustrated using colorimetric maps with five-year intervals to clearly depict changes in incidence over time. We used Microsoft Excel to generate bar graphs for diagnostic stages and number of cases per year over the study period. To describe annual trends, we created line graphs, and logistic regression was used to test the significance of observed trends in HAT incidence at p<0.05.
Ethical considerations: We used routinely-generated program data. Ministry of Health through the VCD gave permission to use the data. The data obtained did not contain personal identifiers. Only the study team had access to the data. Additionally, the US Centers for Disease Control and Prevention (CDC) determined that this study did not qualify as research.
Results


Demographic characteristics and incidence
Overall, there were 2032 HAT cases for the period 2005–2015. The proportion of HAT cases by sub-type was 45% (911/2032)
gambiense HAT cases and 55% (1121/ 2032)
rhodesiense HAT cases. 52% (1056/2032) of cases were males and 48% (976/2032) were females. The median age was 25 years (interquartile range: 14-40) years old. Both
rhodesiense and
gambiense HAT affected males and female in approximately equal proportion: incidence of
gambiense HAT/10,000 was 0.10 among males and 0.091 among females, while incidence of
rhodesiense HAT/10,000 population was 0.76 among males and 0.74 among females. HAT incidence per 10,000 was lowest among children <5 years of age (
gambiense HAT=0.05,
rhodesiense HAT=0.49) and highest among persons >15 years, (
gambiense HAT=0.22,
rhodesiense HAT=2.15) (
Table 1).
Time of diagnosis and treatment outcomes
Overall, 30% (608/2032) of HAT cases were diagnosed in the early stage, 57% (1168/2032) were diagnosed in the late stage, and 13% (255/2032) had unknown time of diagnosis. Among
gambiense HAT patients, 29% (265/911) were diagnosed in the early stage, 44% (398/911) in the late stage, and 27% (247/911) had not been staged. Among
rhodesiense HAT cases, 30% (343/1121) were diagnosed in the early stage, 69% (770/1121) in the late stage and only 0.7% (8/1121) did not record data on disease stage. In all, 97% (247/255) of cases without diagnostic stage data were among
gambiense HAT cases.
Treatment outcomes were categorised as recovered (55%, 1120/2032), died (3%, 67/2032), or unclassified (42%, 845/2032). Among those diagnosed in the early stage, only 1% (6/608) died, while 5% (60/1174) of those who were diagnosed in the late stage died (
Table 2).
Trends in HAT incidence/10,000 population, Uganda, 2005 – 2015
For the period 2005–2015, there was a statistically significant annual decline in
gambiense HAT incidence at a rate of 25% per year (0R=0.75; 95% CI: 0.73-0.77), and
rhodesiense HAT incidence at 13% per year (OR=0.87; 95%CI: 0.85 – 0.89) (
Figure 3).
More than 100 HAT cases were observed during the year 2005 and 2009. There was a sudden drop in HAT cases in 2006 (<55 cases) which was constant for the next three consecutive years (2006–2008). There was a steady decline in number of cases per year after 2009. For every year, there were more than twice the number of late diagnosis patients compared to early diagnosis (
Figure 4).
Rhodesiense HAT was more common in the eastern part of the country and partly in the northern region than
gambiense HAT. Twelve districts of Bugiri, Busia, Dokolo, Iganga, Jinja, Kaberamaido, Kamuli, Lira, Mayuge, Mukono, Soroti, Tororo reported
rhodesiense HAT incidence >0.01 per 10,000 in 2005, thirteen districts of Alebtong, Apac, Budaka, Bugiri, Dokolo, Kaberamaido, Kole, Lira, Namutumba, Palisa, Serere, Soroti, and Wakiso reported
rhodesiense HAT incidence >0.01 in 2010, and four districts of Alebtong, Dokolo, Kaberamaido and Kalangala reported in 2015. Kaberamaido District reported the highest
rhodesiense HAT incidence per 10,000 of 0.12 in 2005 and 0.08 in 2015.
Gambiense HAT was more common in the northwestern part of the country and partly in the northern region. Nine districts of Adjumani, Alebtong, Arua, Dokolo, Kaberamaido, Koboko, Lira, Moyo, and Yumbe reported
gambiense HAT incidence >0.15 per 10,000 in 2005, five districts (Adjumani, Alebtong, Arua, Dokolo, Kaberamaido, and Moyo) reported in 2010, and only one district (Moyo) reported in 2015. Moyo District reported the highest
gambiense HAT incidence per 10,000 of 6.53 in 2005 and 0.097 in 2015. These results show declining incidence of
gambiense HAT over the study period (
Figure 5).
Discussion


Our study reveals that HAT incidence is on the decline in Uganda, and that the targets set by WHO to eliminate HAT as a public health problem are close to being achieved on a district level. The number of endemic districts over this time period has also declined.
Among
rhodesiense HAT cases, most were diagnosed in the late stage. This indicates a missed opportunity for HAT diagnosis. The NSSCP explains the steep drop in the incidence of
rhodesiense HAT in 2006-2008 as being attributed to the massive and active interventions to reduce the tsetse fly population through the use of tsetse fly traps, as well as the mass chemotherapy treatment among the animal (reservoir) population as recommended in previous studies [
9,
21]. The rebound in 2009 was likely due to enhanced surveillance efforts. In 2009,
rhodesiense HAT was spreading rapidly to the northern districts, which were already endemic for
gambiense HAT. Acup
et al in their paper recommended vigorous mechanisms to contain
rhodesiense HAT and limit the infection to the South-Eastern districts, leading to enhanced efforts for disease screening and treatment among human cases in this period [
22].
Many Ugandan families depend on cattle and other livestock as a long-term economic investment. The indigenous cattle are considered by the local population to be disease-resistant and so are not usually treated or sprayed. However, they do harbor trypanosomes, which continuously exposes humans to the disease [
10]. Beyond that, there is continuous buying and selling of cattle between different districts and regions, and this has previously been associated with introduction of trypanosomes in non-endemic districts or geographical locations [
17]. There continues to be increasing scarcity of land available for the growing Ugandan population to use for agriculture and pasture grazing for livestock. This scarcity of land forces livestock farmers to encroach into the tsetse fly breeding zones where they get infected [
23]. During drought, herdsmen have to move long distances in search for pasture and water for their livestock. The movement of cattle during trade and drought to previously non-endemic northern districts in the absence of control measures continues to pose the public health risk of expanded transmission [
8,
10]. Uganda has embraced the “One Health” component as a mechanism to control HAT through vector control [
5], and district authorities from non-endemic areas are implementing the national policy on trypanosomiasis control which demands that all cattle sold should have been treated before sale [
2]. Treating cattle before sale alone requires a lot of veterinary expertise but may help avoid the high costs of managing sleeping sickness patients [
10]. Another study conducted in Uganda, in which village herds of cattle were tested for African Animal Trypanosomiasis (AAT) levels and given mass drug treatment against trypanosomes, showed that the knowledge of livestock AAT levels helps estimate disease burden of
rhodesiense HAT, and mass treatment significantly reduces HAT prevalence in that population [
10].
Males had a higher disease incidence compared to females, and males >15 years were more affected than their female age mates. The higher incidence of HAT among males is likely due to their role as herdsmen/cattle keepers. There were hardly any infants affected by HAT. This agrees with a paper by Andreas et al citing no evidence of mother-to-child transmission; however, this has not been widely studied [
24,
25].
For both HAT sub-types, a larger proportion of HAT cases were diagnosed in the late stage. Literature from Uganda and Kenya reports about 70% of HAT patients are diagnosed and treated wrongly before the correct diagnosis and treatment is commenced [
17]. To some extent, late diagnosis of HAT cases is caused by the consultation with other players in the treatment cascade such as traditional / spiritual healers, which can delay appropriate diagnosis [
17]. In addition, late diagnosis of HAT cases is also likely to be a gap in expertise with HAT diagnosis. Late diagnosis of cases may also be due to a gap in communication or transport systems to bring suspected cases to the SSTC, unclear case definition of what a HAT case is to healthcare workers, lack of technical staff, or insufficient laboratory supplies to facilitate timely diagnosis and later timely referrals. Cases may get misdiagnosed as complicated malaria or meningitis due to the similar central nervous symptomatology [
26].
Limitations
A small proportion of patients had unknown stage of diagnosis, while a significant proportion of patients had unascertained treatment outcome. This could have influenced the findings in either direction, leading to the underestimation or overestimation of late diagnosis counts and poor treatment outcome counts.
Conclusion


Efforts to control acute HAT in Uganda show a positive change towards disease control and the possibilty of elimination, as indicated by declines in disease incidence and mortality. With an annual incidence of <1 HAT case/10,000, Uganda had by 2015 achieved the WHO 2020 elimination target for both rhodesiense and gambiense HAT as a public health threat. However, Uganda has not yet achieved the WHO target for early case detection and diagnosis, as most of the cases are still being diagnosed at the late stage. Authorities in endemic districts should continue routine disease surveillance and screening, early diagnosis, and follow up of all HAT cases to ensure continued success towards elimination
Competing interests


The authors declare no competing interests
Authors´ contributions


KS: Was responsible for the projection conception, and coordination, protocol development, CWW, ET, and AK, who are all placed at the VCD-MOH, participated in data collection, analysis and dissemination as well as manuscript development LB, ETM, and ARA provided technical support during manuscript writing and reviewed the manuscript to ensure intellectual content and scientific integrity.
Acknowledgments


We would like to acknowledge the Uganda Ministry of Health-VCD for allowing us use the routinely generated data. We appreciate Uganda Ministry of Health, AT endemic districts and SSTC staff generating the surveillance data. We also appreciate the African Field Epidemiology Networkl (AFENET) for funding a one week’s scientific writing workshop in which draft 0 of this manuscript was completed. We also appreciate the AFENET Scientific Writers, Dr Baoping Zhu and Dr. Peter Nsubuga for providing technical support during the development of this manuscript. We acknowledge the Uganda Public Health Fellowship Program for providing a platform for scientific writing.
Tables and figures


Table 1: Demographic characteristics and incidence of HAT cases in Uganda, 2005 – 2015, by sub-type
Table 2: Time of diagnosis and treatment outcome of HAT cases, Uganda, 2005 – 2015, by sub-type
Figure 1: Map of Uganda showing the location of Sleeping Sickness Treatment Sites for gambiense HAT and rhodesiense HAT, 2005-2015
Figure 2: Human African Trypanosomiasis surveillance system in Uganda, 2005-2015
Figure 3: Trends in HAT incidence/10,000 population, Uganda, 2005 – 2015
Figure 4: Distribution of rhodesiense HAT cases, by stage of diagnosis, Uganda, 2005 - 2015
Figure 5: Spatial distribution of rhodesiense and gambiense HAT incidence per 10,000 population, 2005, 2010, and 2015
References


- WHO | Trypanosomiasis, human African (sleeping sickness)[Internet]. WHO. [cited 2017 Oct 2].
- Berrang-Ford L, Odiit M, Maiso F, Waltner-Toews D, McDermott J. Sleeping sickness in Uganda: revisiting current and historical distributions. Afr Health Sci. 2006 Dec;6(4):223–31. PubMed | Google Scholar
- Rozendaal JA. Tsetse flies. In: Vector control: methods for use by individuals and communities [Internet]. Geneva: World Health Organization; 1997 [cited 2019 Jul 9]. p. 178–92.
- Wamboga C, Matovu E, Bessell PR, Picado A, Biéler S, Ndung’u JM. Enhanced passive screening and diagnosis for gambiense human African trypanosomiasis in north-western Uganda - Moving towards elimination. PloS One. 2017;12(10):e0186429. https://doi.org/10.1371/journal.pone.0186429 PubMed | Google Scholar
- Zinsstag J, Schelling E, Waltner-Toews D, Whittaker M, Tanner M, C.A.B. International, editors. Human and animal African trypanosomiasis. In: One health: the theory and practice of integrated health approaches [Internet]. Wallingford, Oxfordshire, UK; Boston, MA: CABI; 2015 [cited 2019 Jul 9]. p. 201–21.https://doi.org/10.1079/9781780643410.0000
- WHO | The transmission cycle [Internet]. WHO. [cited 2017 Oct 2].
- Acup C, Bardosh KL, Picozzi K, Waiswa C, Welburn SC. Factors influencing passive surveillance for T. b. rhodesiense human african trypanosomiasis in Uganda. Acta Trop. 2017 Jan;165:230–9.https://doi.org/10.1016/j.actatropica.2016.05.009 PubMed | Google Scholar
- Picozzi K, Fèvre E, Odiit M, Carrington M, Eisler MC, Maudlin I, et al. Sleeping sickness in Uganda: a thin line between two fatal diseases. BMJ. 2005 Nov 24;331(7527):1238–41. https://doi.org/10.1136/bmj.331.7527.1238 PubMed | Google Scholar
- WHO | Eliminating sleeping sickness as a public health problem is on track [Internet]. WHO. [cited 2018 Nov 15].
- Hamill L, Picozzi K, Fyfe J, von Wissmann B, Wastling S, Wardrop N, et al. Evaluating the impact of targeting livestock for the prevention of human and animal trypanosomiasis, at village level, in districts newly affected with T. b. rhodesiense in Uganda. Infect Dis Poverty [Internet]. 2017 [cited 2017 Oct 2];6(1):16.https://doi.org/10.1186/s40249-016-0224-8 PubMed | Google Scholar
- WHO | The Human African Trypanosomiasis Control and Surveillance Programme of the World Health Organization 2000–2009: The Way Forward[Internet]. WHO. [cited 2018 Oct 23].
- WHO | Control and Surveillance [Internet]. WHO. [cited 2018 Oct 23].
- WHO | Eliminating sleeping sickness as a public health problem is on track [Internet]. WHO. [cited 2017 Oct 12].
- UNHCR. Congolese refugees: A protracted situation [Internet]. UNHCR; [cited 2018 Nov 13].
- UNHCR.SOUTH SUDAN SITUATION 2017: Supplementary Appeal January - December 2017 [Internet]. UNHCR; 2017 [cited 2018 Nov 13].
- Palmer JJ, Robert O, Kansiime F. Including refugees in disease elimination: challenges observed from a sleeping sickness programme in Uganda. Conflict and Health. 2017 Dec 1;11(1):22.https://doi.org/10.1186/s13031-017-0125-x PubMed | Google Scholar
- Acup C, Bardosh KL, Picozzi K, Waiswa C, Welburn SC. Factors influencing passive surveillance for T. b. rhodesiense human african trypanosomiasis in Uganda. Acta Tropica. 2017 Jan 1;165:230–9.https://doi.org/10.1016/j.actatropica.2016.05.009 PubMed | Google Scholar
- Uganda Bureau of Statistics. National Population and Housing Census 2014: Provisional Results [Internet]. Kampala; 2014 Nov [cited 2019 Jul 9] p. 1-65.
- Uganda Bureau of Statistics. 2002 Uganda Population and Housing Census: Main Report [Internet]. Kampala, Uganda; 2005 Mar [cited 2019 Jul 9] p. 1–32.
- Uganda Bureau of Statistics.National Population and Housing Census 2014: Main Report [Internet]. Kampala, Uganda; 2016 Mar [cited 2019 Jul 9] p. 1–94.
- Fyfe J, Picozzi K, Waiswa C, Bardosh KL, Welburn SC. Impact of mass chemotherapy in domestic livestock for control of zoonotic T. b. rhodesiense human African trypanosomiasis in Eastern Uganda. Acta Tropica. 2017 Jan 1;165:216–29.https://doi.org/10.1016/j.actatropica.2016.08.022 PubMed | Google Scholar
- Acup, Christine Amongi.Epidemiology and control of human African trypanosomiasis in Uganda [Internet] [Doctoral]. [Edinburgh]: University of Edinburgh; 2014 [cited 2019 Jul 9].
- Wamwiri FN, Changasi RE. Tsetse Flies (Glossina) as Vectors of Human African Trypanosomiasis: A Review. Biomed Res Int. 2016;2016:6201350.https://doi.org/10.1155/2016/6201350 PubMed | Google Scholar
- Lindner AK, Priotto G. The Unknown Risk of Vertical Transmission in Sleeping Sickness—A Literature Review. PLOS Neglected Tropical Diseases. 2010 Dec 21;4(12):e783.https://doi.org/10.1371/journal.pntd.0000783 PubMed | Google Scholar
- Gaillot K, Lauvin M-A, Cottier J-P. Vertical transmission of human African trypanosomiasis: Clinical evolution and brain MRI of a mother and her son. PLOS Neglected Tropical Diseases. 2017 Jul 27;11(7):e0005642. https://doi.org/10.1371/journal.pntd.0005642 PubMed | Google Scholar
- Malvy D, Chappuis F. Sleeping sickness. Clinical Microbiology and Infection. 2011 Jul 1;17(7):986–95.https://doi.org/10.1111/j.1469-0691.2011.03536.x PubMed | Google Scholar