Spatio-temporal distribution of under-five malaria morbidity and mortality hotspots in Ghana, 2012 ŌĆō 2017: a case for evidence-based targeting of malaria interventions
Dora Dadzie1,&, Ernest Kenu1, Nana Yaw Peprah2, Olufemi Olamide Ajumobi3,4, Ben Masiira5, Delia Akosua Bandoh1, Charles Noora Lwanga1, Edwin Afari1
1Ghana Field Epidemiology and Laboratory Training Program Accra, Ghana, 2National Malaria Control Program, Accra, Ghana, 3Nigeria Field Epidemiology and Laboratory Training Program, Abuja, Nigeria, 4African Field Epidemiology Network Nigeria Country Office, Abuja, 5African Field Epidemiology Network Secretariat, Kampala, Uganda
&Corresponding author
Dora Dadzie, Ghana Field Epidemiology and Laboratory Training Program Accra, Ghana
Email: ddadzie8@gmail.com
Introduction:
The spatiotemporal variation in malaria burden underpins the need for targeted malaria interventions. Despite the scale-up of malaria control interventions in Ghana, malaria remains the leading cause of hospital admissions and deaths among children below 5 years (U5). We described spatiotemporal distribution of U5 malaria morbidity and mortality from 2012 to 2017 to provide evidence for deployment of specific malaria interventions to regions of hotspots in Ghana.
Methods:
We conducted a retrospective review of district-level malaria surveillance data from 2012 to 2017. We obtained confirmed U5 malaria case and population data for all districts in Ghana, and computed yearly smoothed malaria incidence and mortality rates. Hotspot analysis was performed using GeoDaŌĆÖs Global and Local Moran I tests of spatial autocorrelation.
Results:
Overall, 8,132,769 U5 malaria cases and 5,932 deaths were reported, with case fatality rate of 0.1%. Under-five malaria incidence increased from 16.4% in 2012 to 31.3% in 2017, and the mortality rate per 100,000 decreased from 30.2 in 2012 to 6.1 in 2017. We found variation in morbidity hotspots from 8 to 23 in the western, south-western and north-eastern areas of the country each year, and six persistent mortality hotspots in the north-eastern areas.
Conclusion:
Over the review period, U5 malaria morbidity increased while mortality decreased. Variability in morbidity hotspots occurred across the western and northern regions unlike persistence of mortality hotspots in the north-eastern region. We recommend that the National Malaria Control Program systematically deploys preventive and case management interventions to areas of hotspots and also conduct a further evaluation to identify the causes of high mortality in the northeastern areas.
Introduction

Globally, 216 million malaria cases and 445,000 associated deaths were reported in 2016. Sub-Saharan Africa contributed more than 90% of cases and deaths [1]. In the early 2000s, the global goal to halt malaria and reverse its incidence prompted additional investment in malaria control [1]. An evaluation conducted 15 years later revealed a global decline of malaria morbidity by 37% and mortality by 60% [2]. In Ghana, malaria prevalence in children under five years (U5) decreased from 75% in 2000 to 21% in 2016, while mortality declined by 60% from 2000 to 2016 [3,4]. Between 2010 and 2017 however, the prevalence stalled at 21% to 28% despite scale-up of interventions [5,6]. In some regions, U5 malaria prevalence increased during this period [7,8]. So, by 2016, malaria remained the leading cause of U5 hospital admissions and mortality in Ghana. In 2016, 182,438 U5 malaria cases (i.e. 48% of U5 hospital admissions) and 590 U5 deaths (i.e. 46.7% of U5 deaths) were reported by the malaria surveillance system in Ghana [7].
┬Ā
The National Malaria Control Program in Ghana targets to reduce malaria morbidity and mortality by 75% from 2012 levels by 2020 [
4]. The estimated funds required to achieve this target is over USD 2.2 billion. However, available funding falls short by USD 854 million [
4]. It has, therefore, become necessary that the program adopts approaches that would optimise the impact of control measures and accelerate progress towards the set targets. Malaria morbidity and mortality exhibit variations across locations and time, which are essential for allocating resources for malaria control [
9ŌĆō11]. When resource allocation is not targeted, high-risk locations may not receive quantities of resources proportional to their risk, therefore have sustained high risk [
9,
12,
13]. Malaria prevalence surveys in Ghana demonstrate regional differences in malaria endemicity, and these have guided some interventions previously [
5,
6]. However, even within regions, malaria is unevenly distributed, suggesting that reliance on regional variations to target interventions is not sufficient [
14,
15].
┬Ā
To optimise the impact of control measures, stratification of malaria intensity at the lowest possible sub-national levels, identification of hotspots, and devotion of more resources to these locations have been recommended [
16]. Malaria hotspots describe foci of high malaria risk or increased risk of malaria emergence [
17,
18]. In other African countries, malaria hotspots detection to guide malaria control and monitor intervention coverage have been implemented [
19ŌĆō24]. In Ghana, earlier attempts to map hotspots were limited by inadequate malaria data at sub-national levels or did not consider the temporal dynamics of hotspots [
25,
26]. Thus, available maps do not provide sufficient information on malaria hotspots, needed to guide targeting of interventions at sub-national levels. We described the spatiotemporal distribution of U5 malaria morbidity and mortality hotspots from 2012 to 2017 to provide information for targeting of malaria interventions.
Methods


Study area
┬Ā
Ghana is a West African country located between longitudes 4 ┬░ W and 2 ┬░ E, and latitudes 4 ┬░ N and 12 ┬░ N. The country has a U5 population of 5,362,184 (projected from 2010 census). There are ten regions, which are further divided into 216 districts. The median district population size is 19,532 (range: 4,543 to 371,748). Malaria is endemic in all regions with a national U5
P. falciparum prevalence of 21% [
5]. The regional prevalences demonstrate varying levels of malaria endemicity with only Greater Accra (5%) having low malaria transmission intensity. The remaining nine regions have moderate malaria transmission intensities and range from 15% to 31% [
5]. The terrain of Ghana is mostly low lying with highlands in the middle belt. The tropical climate in Ghana supports malaria transmission all year round with seasonal peaks during the rainy season (June - October). The northern belt of the country experiences six months of rainy season, the middle, seven months of rainy season, and the coastal areas, five months of rainy season. Average day and night temperatures are 30 Ōäā and 24 Ōäā respectively, with temperatures comparatively higher in the northern belt, than middle and southern belts. The geographic variations in climate and ecology form the basis for the malaria ecologic zones. The guinea savannah occupies the northern belt and expresses the highest malaria risk. The rainforest, which occupies the middle belt has the second highest malaria risk, and, the coastal and mangrove swamp, found along the coastline has the least malaria risk.
┬Ā
Surveillance system for malaria
┬Ā
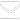
The malaria surveillance system in Ghana is passive. Malaria is confirmed by a parasitological diagnosis of a febrile case harbouring
Plasmodium parasites with microscopy or malaria rapid diagnostic test. Cases and deaths are reported on a monthly basis as aggregates by health facilities through the District Health Information Management System (DHIMS2). The DHIMS2 is an electronic data collection tool that captures data from all health facilities at the community level. Data aggregation is performed at the facility, district, regional and national levels (
Figure 1).
┬Ā
Study design
┬Ā
We conducted a retrospective review of district-level malaria surveillance data from 2012 to 2017. These were further analysed and described spatially.
┬Ā
Data extraction
┬Ā
We extracted confirmed U5 malaria cases for 2012 to 2017 from DHIMS2. Variables extracted from DHIMS2 were U5 uncomplicated malaria cases, U5 severe malaria cases, U5 malaria deaths, and completeness of malaria reporting by health facilities. Completeness of reporting was defined as the proportion of expected reports from health facilities that were submitted. DistrictsŌĆÖ population data were obtained from the Ghana Statistical Service (GSS). Malaria data from all 216 districts in Ghana were used for analysis.
┬Ā
Data processing and analysis
┬Ā
Surveillance and population data were matched to corresponding districts on a district boundary map of Ghana using GeoDa version 1.8 [
27], a geographical information system (GIS) software. An exploratory analysis using filters and counts was performed to identify missing and invalid records, which were not included in further analysis. We estimated the unit effect of increasing completeness of reporting on U5 malaria incidence using simple linear regression in Stata version 14. We used completeness of reporting as the predictor variable and U5 malaria incidence as outcome variable. A
p-value of 0.05 indicated a significant effect of completeness of reporting on U5 malaria incidence. The estimation was done to provide a context to interpret findings.
┬Ā
Descriptive statistics
┬Ā
We computed frequencies of cases and deaths, the proportion of severe cases, case fatality rates, malaria-specific mortality rates, and medians and interquartile ranges (IQR) of variables in STATA version 14. Yearly mortality rates were computed using U5 malaria deaths as numerator and mid-year U5 population as the denominator. We determined the monthly distribution of U5 malaria cases.
┬Ā
Empirical Bayes smoothing
┬Ā
We imported the spatially-linked case and population-at-risk data into GeoDa. The yearly cumulative incidence of malaria was calculated for each district from 2012 to 2017 using Empirical Bayes Smoothing (EBS) approach. In mapping, crude rates across spatial units with different population sizes are not comparable due to differences in their standard errors and consequently precision of crude rates. The need for smoothing arose because units with smaller populations tend to have higher standard errors and therefore less precise rates, compared to units with larger population sizes [
28]. To adjust for the differences in the districtsŌĆÖ population sizes and improve the precision of incidence, we used the EBS function in GeoDa to compute smoothed U5 malaria incidence. The EBS incidence is a weighted average of the crude incidence and national average. For each district ŌĆśiŌĆÖ, the smoothed U5 malaria incidence is given by;

is the crude incidence of district ŌĆśiŌĆÖ, given by the number of U5 malaria cases divided by the U5 population at risk,
╬Ė is the national average,
Žā is the variance of the prior distribution,
╬╝ is mean of the prior distribution, and
Pi is the population at risk in district ŌĆśiŌĆÖ.
Žā and
╬╝ are estimated from the data [
28].
┬Ā
We drew choropleth maps using the smoothed incidence. We categorised districts into four classes of malaria transmission intensity, per World Health Organisation (WHO) recommendation [
29]. These groups were; high malaria transmission intensity ( Ōēź 45%), moderate malaria transmission intensity (25% - 45%), low malaria transmission intensity (10% - 25%), and very low malaria transmission intensity < 10% [
29].
┬Ā
Global Moran I
┬Ā
We measured Global Moran I to evaluate overall spatial autocorrelation of U5 malaria morbidity and mortality in GeoDa [
27]. This function measured how districts were similar to nearby districts (neighbours) concerning U5 malaria morbidity and mortality. We defined neighbours by first-order queen contiguity spatial weight matrix, which indicated that only districts sharing common borders were considered as neighbours. We reported only positive Moran I values, with significant p-values due to the interest in identifying only hotspots. A positive Moran I value with
p-value < 0.05, however, indicated spatial clustering of high incidence districts or low incidence districts. The degree of clustering was determined by the proximity of Moran I value to one, with values approaching one showing strong clustering, and those approaching zero, showing weak or no spatial clustering.
┬Ā
Local Moran I
┬Ā
Global Moran I assessed overall spatial autocorrelation without pinpointing the locations of clusters. We identified the location of hotspots using GeoDaŌĆÖs Local Indicator of Spatial Autocorrelation (LISA) [
27]. We defined malaria morbidity hotspots and mortality hotspots as high-risk clusters for malaria morbidity and mortality respectively. The clusters denoted districts with significantly higher than expected concentration of malaria morbidity or mortality. We presented only significant high-risk clusters on GIS-generated cluster maps.
┬Ā
Ethical considerations
┬Ā
Secondary data were used for the study. Therefore no ethical approval was required. However, we obtained approval from the Ghana Health Service Ethical Review Board with identification number GHS-ERC155/12/17ID. Permission was also obtained from the National Malaria Control Program and Ghana Statistical Service to use their data. The data used did not contain any personal identifiers.
Results


Completeness of malaria reporting
┬Ā
Completeness of malaria reporting by all health facilities increased from 62.1% in 2013 to 92.8% in 2017 (
Table 1). The median completeness of reporting by district was 64.2% (IQR: 48 - 77) in 2013, 69.4% (IQR: 53.5 ŌĆō 82.5) in 2014, 75.5% (IQR: 58.8 ŌĆō 87.0) in 2015, 84.0% (IQR: 70.4 ŌĆō 93.6) in 2016, and 96.3% (IQR: 90.0 ŌĆō 98.9) in 2017. A percent increase in completeness of reporting by district was estimated to significantly increase malaria incidence by 0.003 (R
2 = 4.2%,
p-value < 0.01) in 2016 and 0.005 (R
2 = 2.7%,
p-value = 0.02) in 2017. No significant relationship was found between completeness of malaria reporting and U5 malaria incidence in 2013 to 2015 (all
p-values > 0.05).
┬Ā
Incidence and mortality rates
┬Ā
From 2012 to 2017, the malaria surveillance system recorded 8,132,769 confirmed U5 malaria cases and 5,932 deaths, with case fatality rate (CFR) of 0.1%. Severe malaria cases were 4.9% (400,436 / 8,132,769). Under-five malaria incidence increased from 16.4% in 2012 to 31.3% in 2017, and malaria-specific mortality rate decreased from 30.2/100,000 in 2012 to 6.1/100,000 in 2017 (
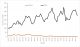
Table 1). For all years, U5 malaria incidence began to rise in April, peaked twice, in July and October-November, before declining in December. Severe malaria incidence decreased over the period under study (
Figure 2).
┬Ā
Spatial characteristics
┬Ā
The incidence of U5 malaria increased from 2012 to 2017 for about 99% of districts. The medians for yearly U5 malaria incidence were 16.7% (IQR: 8.9 - 27.6) in 2012, 24.6% (IQR: 16.2 - 39.2) in 2013, 29.0% (IQR: 18.7 - 44.6) in 2014, 37.7% (IQR: 23.1 - 50.8) in 2015, 35.4% (IQR: 23.6 - 52.07) in 2016, and 36.0% (IQR: 23.5 - 53.3) in 2017. The number of districts with high transmission intensity increased from 17 in 2012 to 77 in 2017. These districts were in mostly western, south-western and north-eastern parts of the country (
Figure 3).
The highest Moran I value for morbidity was observed in 2014 and 2017 (Moran I of 0.5, p < 0.05), and lowest in 2012 (Moran I of 0.1,
p < 0.05). Under-five malaria mortality exhibited less spatial clustering than U5 morbidity (
Table 2).
┬Ā
Between 8 and 23 significant hotspots of malaria morbidity were detected each year, with more hotspots observed in 2015 - 2017. The hotspots were located in the northern belt in 2013 and 2014. From 2015 through 2017, more hotspots were found in the western and south-western parts of the country. Three hotspots for malaria morbidity persisted from 2012 to 2017 (
Figure 3). Six hotspots of U5 malaria mortality persisted in the north-eastern part of the country. Few districts were hotspots of both morbidity and mortality (
Figure 3).
Discussion


We found evidence of significant spatial clustering of U5 malaria morbidity and mortality in Ghana from 2012 to 2017. Hotspots of U5 malaria morbidity persisted in the western and south-western parts of the country, while those of mortality persisted mainly in the north-eastern part. Also in the north-eastern part, morbidity hotspots were present in 2012, but many had disappeared by 2017.
┬Ā
The National Malaria Control Programme employs three main approaches to malaria control; integrated vector control (mainly use of insecticide-treated net [ITN], indoor residual spraying [IRS] and larviciding), chemoprevention, and early case diagnosis and management [
5,
14]. Since 2010, there have been three rounds of mass ITN distribution (in 2012, 2014 and 2017), and these have been supplemented with continuous ITN distribution through antenatal clinics, child welfare clinics and primary schools [
5]. Unlike ITNs, IRS and larviciding have been limited to few districts due to their associated high cost. Larviciding has only been used in few urban districts in the south, while IRS and seasonal malaria chemoprevention (SMC) have been used selectively in the north [
14,
30]. The apparent migration of morbidity hotspots from the north towards the south may be explained by the particular focus of the malaria programme in the northern regions. Over the years, the unique malaria epidemiology (i.e., highest malaria burden) of northern regions informed the implementation of targeted interventions SMC and IRS in addition to ITN in these regions [
5,
14,
30]. The disappearance of hotspots in these regions over the years reflects the effectiveness of these interventions and highlight the importance of a targeted approach to the deployment of interventions. The persistence of morbidity hotspots in five districts in the north-eastern part despite being beneficiaries of the additional interventions remains unclear.
┬Ā
The distribution of morbidity hotspots shown in our risk maps is strikingly similar to malaria risk distribution predicted by Kumi-Boateng
et al. and the malaria control programme. In the study by Kumi-Boateng
et al., which used only climatic and environmental factors for malaria risk prediction, the south-western and north-eastern parts of Ghana were shown to have the highest malaria risk. Also, a study by the malaria control programme revealed that the south-western and north-eastern parts of the country have the highest U5 malaria prevalence [
25].
┬Ā
In our study, we found out that, some districts in the north-eastern part which had relatively lower U5 malaria incidence were hotspots for U5 malaria mortality. The northern part of Ghana has the highest proportion of residents living in poverty [
31]. Previous studies done in Ghana and Tanzania indicate that children living in poor areas have greater risk of death due to associated barriers in access to healthcare [
32,
33]. In another study by Fobil
et al., low socioeconomic status was associated with higher malaria mortality [
34]. In the northern part, health facilities are few, sparsely distributed in the communities. Thus there is limited access of the population to needed health care [
31,
35]. Children below five years who are missed by the key interventions and fall ill from malaria are likely to present in the later stage of malaria, and are prone to a higher risk of dying. Thus, although the interventions may be working to reduce morbidity, mortality is still a high probability for children missed by the health system.
┬Ā
We also found U5 malaria incidence to increase while severe malaria incidence and mortality declined across the country. This increase in U5 malaria incidence is unanticipated given the scale-up of malaria interventions in recent years. The finding of a slightly increasing trend for malaria incidence may be attributed to a number of reasons. In this study, only confirmed malaria cases were analysed. The introduction of the ŌĆśtest treat and track policyŌĆÖ for suspected malaria in 2012, with a corresponding improvement in confirmatory malaria diagnosis, may account for the observed increase in confirmed malaria cases over the years [
7]. Completeness of reporting increased from 62% in 2013 to 93% in 2017.
┬Ā
Nevertheless, the estimated effect of this increase on malaria incidence was found to be small. This finding is not surprising because records from DHIMS2 show that government facilities contribute 62% of U5 malaria cases, faith-based facilities, 15%, and private hospitals, 24%. These health facilities have persistently reported since 2013, with other private health facilities contributing less than 4% to the increase over the years [
36]. Thus, the increase in the completeness of reporting may not be contributing significantly to the rising confirmed malaria incidence or may be contributing smaller increases than assumed The decline in severe malaria and mortality could be due to improvement in early malaria case diagnosis and management. Death is the end of spectrum of severe malaria clinical syndrome, preventable by timely case management. In two studies in Rwanda and Ethiopia, increase in the availability of antimalarial drugs was linked to reductions in U5 malaria deaths by more than 50% [
37,
38].
┬Ā
We found U5 malaria incidence to be unevenly distributed over the months with more cases recorded from June to November in comparison with December to May. This distribution corroborates what has been documented in earlier studies and follows a pattern similar to the monthly distribution of rainfall, with the bimodal malaria peaks coinciding with rainfall peaks [
39ŌĆō41].
┬Ā
The study has some limitations. Routine surveillance data from health facilities were analyzed, and findings are prone to underestimation. Completeness of reporting by districts varied. However, this did not significantly influence districtsŌĆÖ malaria incidence except in 2015 - 2017, where 1% increase in the completeness of reporting was estimated to increase malaria incidence by only 0.002 - 0.005. Only confirmed cases were used for analysis. Thus, the possibility of excluding cases that were not tested but could otherwise have been positive, cannot be ruled out. Malaria case-patients may have accessed health facilities outside of their districts, thus captured and recorded in a different district. Moreover, these were most probably for extremely few cases and could not have had an impact on the number of cases analysed. Despite these limitations, findings from this study are still useful for spatial targeting of malaria interventions.
Conclusion


We identified stable U5 malaria morbidity and mortality hotspots in Ghana from 2012 to 2017. Morbidity hotspots were located mostly in the western, south-western and north-eastern parts, while mortality hotspots were located in the north-eastern part. Malaria in children less than five years increased while severe malaria, and malaria mortality decreased over the period under study. Based on our findings, we recommend that the National Malaria Control Programme should evaluate malaria interventions and devote critical effective malaria prevention interventions to the south-western, western and north-eastern parts of the country. Further studies are required to assess malaria case management, and to identify the causes of high mortality in the north-eastern areas. The public health directorates should also intensity malaria prevention education in south-western, western and north-eastern parts of the country.
Competing interests


The authors declare that they have no competing interests.
Funding
┬Ā
The study was funded by President Malaria Initiative (PMI).
AuthorsŌĆÖ contributions


DD and EK conceptualised the study and designed the study protocol. DD was responsible for data collection, cleaning, analysis and interpretation. EK contributed to data analysis and interpretation. NYP, OOA, BM, DAB, CNL and EA contributed to data interpretation and revised the manuscript for intellectual content. All authors read and approved the final manuscript.
Acknowledgements


The authors are grateful to PMI and staff of National Malaria Control Program, Accra, Ghana.
Tables and figures


Table 1: Distribution of under-five malaria incidence, case fatality rates, malaria-specific mortality rates and completeness of malaria reporting by health facilities, Ghana, 2012 ŌĆō 2017
Table 2: Global Moran I results and p-values for under-five morbidity and mortality, Ghana, 2012 ŌĆō 2017
Figure 1: Information flow chart for Malaria surveillance system, Ghana
Figure 2: Monthly distribution of under-five malaria incidence and severe malaria incidence, Ghana, 2012 ŌĆō2017
Figure 3: Spatial distribution of under-five malaria morbidity, Ghana, 2012 ŌĆō 2017
Figure 4: Spatial hotspots of under-five malaria morbidity and mortality, Ghana, 2012 ŌĆō 2017
References


- World Health Organization. World malaria report 2017 [Internet]. Geneva; 2017 [cited 2019 Jul 23] p. 196.
- World Health Organization .World malaria report 2016 [Internet]. Geneva; 2016 [cited 2019 Jul 23] p. 186.
- Awine T, Malm K, Bart-Plange C, Silal SP. Towards malaria control and elimination in Ghana: challenges and decision making tools to guide planning. Glob Health Action. 2017 Jan;10(1):1381471.https://doi.org/10.1080/16549716.2017.1381471 PubMed | Google Scholar
- National Malaria Control Programme (NMCP). Strategic Plan for Malaria Control in Ghana 2014ŌĆō 2020. 201.
- Ghana Statistical Service (GSS), Ghana Health Service (GHS), ICF.Ghana Malaria Indicator Survey 2016 [Internet]. Accra, Ghana, and Rockville, Maryland, USA; 2017 May [cited 2019 Jul 23] p. 138.
- Ghana Statistical Service. Ghana Multiple Indicator Cluster Survey with an Enhanced Malaria Module and Biomarker. Final Rep. 2011;1ŌĆō450.
- Ghana Health Service. Ghana National Malaria Control Programme Annual Bulletin. 2016. Report No.: 104.
- Ghana Health Service. 2015 Annual Report National Malaria Control Programme. 2016 p. 1ŌĆō70.
- Bousema T, Griffin JT, Sauerwein RW, Smith DL, Churcher TS, Takken W, et al. Hitting Hotspots: Spatial Targeting of Malaria for Control and Elimination. PLoS Med. 2012 Jan 31;9(1):e1001165.https://doi.org/10.1371/journal.pmed.1001165 PubMed | Google Scholar
- Bousema T, Drakeley C, Gesase S, Hashim R, Magesa S, Mosha F, et al. Identification of Hot Spots of Malaria Transmission for Targeted Malaria Control. J Infect Dis. 2010 Jun;201(11):1764ŌĆō74.https://doi.org/10.1086/652456 PubMed | Google Scholar
- Srivastava A, Nagpal BN, Joshi PL, Paliwal JC, Dash AP. Identification of malaria hot spots for focused intervention in tribal state of India: a GIS-based approach. Int J Health Geogr. 2009;8(1):30.https://doi.org/10.1186/1476-072X-8-30 PubMed | Google Scholar
- Diboulo E, Si├® A, Vounatsou P. Assessing the effects of malaria interventions on the geographical distribution of parasitaemia risk in Burkina Faso. Malar J. 2016 Dec;15(1):228.https://doi.org/10.1186/s12936-016-1282-x PubMed | Google Scholar
- Shirayama Y, Phompida S, Shibuya K. Geographic information system (GIS) maps and malaria control monitoring: intervention coverage and health outcome in distal villages of Khammouane province, Laos. Malar J. 2009;8(1):217.https://doi.org/10.1186/1475-2875-8-217 PubMed | Google Scholar
- JSI Research & Training Institute, Inc.Report of the Ghana Urban Malaria study [Internet]. Accra, Ghana; 2013 Jan [cited 2019 Jul 23] p. 112.
- Awine T, Malm K, Peprah NY, Silal SP. Spatio-temporal heterogeneity of malaria morbidity in Ghana: Analysis of routine health facility data. Munderloh UG, editor. PLOS ONE. 2018 Jan 29;13(1):e0191707.https://doi.org/10.1371/journal.pone.0191707 PubMed | Google Scholar
- World Health Organization, Global Malaria Programme, World Health Organization. A framework for malaria elimination [Internet]. 2017 [cited 2019 Jul 23].
- Ernst KC, Adoka SO, Kowuor DO, Wilson ML, John CC. Malaria hotspot areas in a highland Kenya site are consistent in epidemic and non-epidemic years and are associated with ecological factors. Malar J. 2006 Sep 13;5:78.https://doi.org/10.1186/1475-2875-5-78 PubMed | Google Scholar
- McKay HS, Lessler J, Moore SM, Azman AS. What is a Hotspot Anyway? Am J Trop Med Hyg. 2017 Jun 7;96(6):1270ŌĆō3.https://doi.org/10.4269/ajtmh.16-0427 Google Scholar
- Yeshiwondim AK, Gopal S, Hailemariam AT, Dengela DO, Patel HP. Spatial analysis of malaria incidence at the village level in areas with unstable transmission in Ethiopia. Int J Health Geogr. 2009;8(1):5.https://doi.org/10.1186/1476-072X-8-5 PubMed | Google Scholar
- Bejon P, Williams TN, Liljander A, Noor AM, Wambua J, Ogada E, et al. Stable and Unstable Malaria Hotspots in Longitudinal Cohort Studies in Kenya. Smith TA, editor. PLoS Med. 2010 Jul 6;7(7):e1000304.https://doi.org/10.1371/journal.pmed.1000304 PubMed | Google Scholar
- Platt A, Obala AA, MacIntyre C, Otsyula B, Meara WPO. Dynamic malaria hotspots in an open cohort in western Kenya. Sci Rep. 2018 Dec;8(1):647.https://doi.org/10.1038/s41598-017-13801-6 PubMed | Google Scholar
- Ouedraogo B, Inoue Y, Kambir├® A, Sallah K, Dieng S, Tine R, et al. Spatio-temporal dynamic of malaria in Ouagadougou, Burkina Faso, 2011ŌĆō2015. Malar J. 2018 Dec;17(1):138.https://doi.org/10.1186/s12936-018-2280-y PubMed | Google Scholar
- Kabaghe AN, Chipeta MG, McCann RS, Phiri KS, van Vugt M, Takken W, et al. Adaptive geostatistical sampling enables efficient identification of malaria hotspots in repeated cross-sectional surveys in rural Malawi. Yukich J, editor. PLOS ONE. 2017 Feb 14;12(2):e0172266. https://doi.org/10.1371/journal.pone.0172266 PubMed | Google Scholar
- Mwakalinga VM, Sartorius BKD, Mlacha YP, Msellemu DF, Limwagu AJ, Mageni ZD, et al. Spatially aggregated clusters and scattered smaller loci of elevated malaria vector density and human infection prevalence in urban Dar es Salaam, Tanzania. Malar J. 2016 Dec;15(1):135.https://doi.org/10.1186/s12936-016-1186-9 PubMed | Google Scholar
- National Malaria Control Programme, University of Health & Allied Sciences, AGA Malaria Control Programme, World Health Organization, INFORM Project. An epidemiological profile of malaria and its control in Ghana. A report prepared for the Ministry of Health, the Roll Back Malaria Partnership and the Department for International Development, UK. 2013;(November).
- Kumi-Boateng B, Stemn E, Mireku-Gyimah Kumi-Boateng D. Modelling of Malaria Risk Areas in Ghana by using Environmental and Anthropogenic Variables ŌĆō A Spatial Multi- Criteria Approach*. Ghana Min J. 2015;15(2):1ŌĆō10. PubMed | Google Scholar
- Anselin L, Ibnu S, Youngihn K. GeoDa: An Introduction to Spatial Data Analysis. Geographical Analysis. 2006;38(1):5ŌĆō22.https://doi.org/10.1111/j.0016-7363.2005.00671.x PubMed | Google Scholar
- Anselin L, Lozano N, Koschinsky J. Rate transformations and smoothing. Urbana. 2006;51:61801. PubMed | Google Scholar
- World Health Organization, Global Malaria Programme, World Health Organization. A framework for malaria elimination [Internet]. 2017 [cited 2019 Jul 23].
- Ghana Health Service. 2015 Annual Report: National Malaria Control Programme [Internet]. Accra, Ghana; 2016 Feb [cited 2019 Jul 23] p. 70.
- Cooke E, Hague S, McKay A. The Ghana Poverty and Inequality Report ŌĆō 2016. UNICEF. 2016;1ŌĆō43.
- Selemani M, Mrema S, Shamte A, Shabani J, Mahande MJ, Yeates K, et al. Spatial and spaceŌĆōtime clustering of mortality due to malaria in rural Tanzania: evidence from Ifakara and Rufiji Health and Demographic Surveillance System sites. Malar J. 2015 Dec;14(1):369. https://doi.org/10.1186/s12936-015-0905-y PubMed | Google Scholar
- Lartey ST, Khanam R, Takahashi S. The impact of household wealth on child survival in Ghana. J Health Popul Nutr. 2016 22;35(1):38.https://doi.org/10.1186/s41043-016-0074-9 PubMed | Google Scholar
- Fobil JN, Levers C, Lakes T, Loag W, Kraemer A, May J. Mapping Urban Malaria and Diarrhea Mortality in Accra, Ghana: Evidence of Vulnerabilities and Implications for Urban Health Policy. J Urban Health. 2012 Dec;89(6):977ŌĆō91.https://doi.org/10.1007/s11524-012-9702-x PubMed | Google Scholar
- Ghana Statistical Service. Ghana Statistical Living Standards Survey 6 Poverty Profile. J Chem Inf Model. 2013;53(9):1689ŌĆō1699. PubMed | Google Scholar
- Ghana Ministry of Health. Centre for Health Information Management / District Health Information Management System Version 2.9, 2018.
- Karema C, Aregawi MW, Rukundo A, Kabayiza A, Mulindahabi M, Fall IS, et al. Trends in malaria cases, hospital admissions and deaths following scale-up of anti-malarial interventions, 2000ŌĆō2010, Rwanda. Malar J. 2012;11(1):236.https://doi.org/10.1186/1475-2875-11-236 PubMed | Google Scholar
- Aregawi M, Lynch M, Bekele W, Kebede H, Jima D, Taffese HS, et al. Time Series Analysis of Trends in Malaria Cases and Deaths at Hospitals and the Effect of Antimalarial Interventions, 2001ŌĆō2011, Ethiopia. Lubell Y, editor. PLoS ONE. 2014 Nov 18;9(11):e106359.https://doi.org/10.1371/journal.pone.0106359 PubMed | Google Scholar
- Dery DB, Brown C, Asante KP, Adams M, Dosoo D, Amenga-Etego S, et al. Patterns and seasonality of malaria transmission in the forest-savannah transitional zones of Ghana. Malar J. 2010;9(1):314.https://doi.org/10.1186/1475-2875-9-314 PubMed | Google Scholar
- Lacombe G, McCartney M, Forkuor G. Drying climate in Ghana over the period 1960ŌĆō2005: evidence from the resampling-based Mann-Kendall test at local and regional levels. Hydrol Sci J. 2012;57(8):1594ŌĆō609.https://doi.org/10.1080/02626667.2012.728291 PubMed | Google Scholar
- Nkrumah F, Klutse NAB, Adukpo DC, Owusu K, Quagraine KA, Owusu A, et al. Rainfall Variability over Ghana: Model versus Rain Gauge Observation. Int J Geosci. 2014;05(07):673ŌĆō83.https://doi.org/10.4236/ijg.2014.57060 PubMed | Google Scholar